Investigation of Pore Size Particle Size and Column Geometry Effects on SEC Analysis of Monoclonal Antibodies
Smaller particle size stationary phases have seen increased use in the modern analytical laboratory due to their ability to exhibit greater resolution while enabling the use of smaller column geometries. Shorter column lengths and narrower bores allow for faster run times and lower flow rates, decreasing laboratory costs in terms of solvent consumption and man-hours needed to run the analyses. This application note examines the effects of smaller particle sizes in various column geometries on size-exclusion chromatography (SEC) methods and the method variables affecting the characterization of antibody products.
Smaller particle size stationary phases have seen increased use in the modern analytical laboratory due to their ability to exhibit greater resolution while enabling the use of smaller column geometries. Shorter column lengths and narrower bores allow for faster run times and lower flow rates, decreasing laboratory costs in terms of solvent consumption and man-hours needed to run the analyses. This application note examines the effects of smaller particle sizes in various column geometries on size-exclusion chromatography (SEC) methods and the method variables affecting the characterization of antibody products.

This work focused on scaling-down a monoclonal antibody SEC method, starting with a typical analytical-size SEC column (YMC-Pack Diol 5 µm, 300 Å, 300 ??8.0 mm) and ending with a small particle, shorter length, narrower-bore column (YMC-pack Diol 2 µm, 300 Å, 150 ??4.6 mm). The purpose of this experiment was to detail the differing performance characteristics exhibited by each column as physical properties were altered, one variable at a time.

Operating Parameters
Sample: 1 mg/mL Avastin in PBS
Mobile Phase: 100 mM sodium phosphate with
200 mM sodium chloride at pH=7.0
Column Temp: 25 °C
Flowrate: 1.0 mL/min for 8.0 mm I.D columns
0.33 mL/min for 4.6 mm I.D. columns
Inj. Volume: 30 µL for 8.0 mm I.D. columns
10 µL for 4.6 mm I.D. columns
HPLC System: Agilent 1260
Changes in column geometry were addressed by scaling flow rate (1.0 mL/min to 0.33 mL/min) and injection volume (30 µL to 10 µL) accordingly as inner diameter was decreased from 8.0 mm to 4.6 mm. This allows for a method that is typically run at 1.0 mL/min for 15 min to be run at 0.33 mL/min for 10 min, cutting run time by 1/2 and solvent consumption by a factor of 6x. For resolution comparability see Table I.
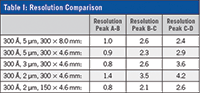
As the results indicate, when applied to SEC analysis of monoclonal antibodies, smaller particle stationary phases in shorter columns can increase throughput, while still providing adequate resolution that rivals the standard 300 ? 8.0 mm, 5 µm columns.

YMC America, Inc.
941 Marcon Blvd., Suite 201, Allentown, PA 18109
tel. (610) 266-8650, fax (610) 266-8652
Website: ymcamerica.com

Analysis of Greenhouse Gases by Gas Chromatography
May 15th 2024This application note demonstrates the use of SCION's 8500GC system for the analysis of key greenhouse gases—carbon dioxide, methane, and nitrous oxide—in a single atmospheric air matrix. Highlighting the system's excellent sensitivity and repeatability, this method is crucial for understanding emission sources and combating climate change.
Analysis of DEHP in Drinking Water by HPLC-DAD
May 15th 2024This application note outlines a method for detecting low levels of 1,4-Dioxane, a likely carcinogenic compound, in drinking water using SCION GC-MS technology. It details the procedure for analyzing water samples spiked with 1,4-Dioxane, achieving excellent sensitivity and low detection limits, demonstrating the method's effectiveness for environmental monitoring.
Plant Protection Product Impurity Screening by GC-FID with GC-MS Confirmation
May 15th 2024This application note from SCION Instruments delves into the meticulous screening of impurities in plant protection products using GC-FID, with GC-MS for confirmation. It emphasizes the necessity of identifying and quantifying impurities to comply with regulatory standards in industries such as pharmaceuticals, food, and agriculture. The study specifically examines eugenol, showcasing how to determine significant impurities for regulatory submission. The approach integrates GC-MS and analytical standards, ensuring precise impurity identification and quantification critical for product approval.