Hamilton PRP-C18 High-Efficiency Separations at Any pH
Special Issues
The Hamilton PRP-C18 is a column designed to provide high-efficiency reversed phase separations over an extended column life in nearly any mobile phase or pH. The rigid stationary phase has excellent mechanical and thermal stability (> 100 °C), does not experience shrinkage or swelling and is completely inert to most conditions commonly encountered in reversed phase chromatography. In this study the pH stability of the PRP-C18 is evaluated. Even after prolonged exposure to concentrated (1 molar) NaOH and H2SO4 there was no measurable deterioration in performance.
Octadecyl silane (ODS) is the prevailing stationary phase in reversed-phase HPLC. Despite a well-celebrated, widespread use in chromatography, traditional ODS columns are not without limitations. Acidic conditions promote hydrolytic stripping of octadecyl functionalization, while alkaline conditions (pH > 7) attack the silica bed, both of which are principle sources for anomalous peak shape and shifting retention times that progressively worsen over the life of the column.
The PRP-C18 reversed phase column has similar mechanical stability (up to 5000 psi) and separation efficiency to that of traditional ODS, but without many of the chemical restrictions. The PRP-C18 stationary phase does not experience stripping or dissolution under the most extreme conditions. This allows for an expanded mobile phase repertoire for use in methods development or aggressive regeneration procedures.
Experimental Conditions
Column: PRP-C18, 4.1 × 50 mm, 5 µm
Instrumentation: Agilent 1100 quaternary pump with UV detector
Standards: acetone, phenol, benzyl alcohol, benzene, toluene, ethylbenzene, propylbenzene, napthalene, pentylbenzene, hexylbenzene
Mobile phase A: 0.2% Phosphoric acid
Mobile phase B: A + 95% ACN
Gradient: 5 to 100% B in 20 min
Flow rate: 2 mL/min
Temperature: Ambient
Injection volume: 2 µL
Detection: UV at 205 nm
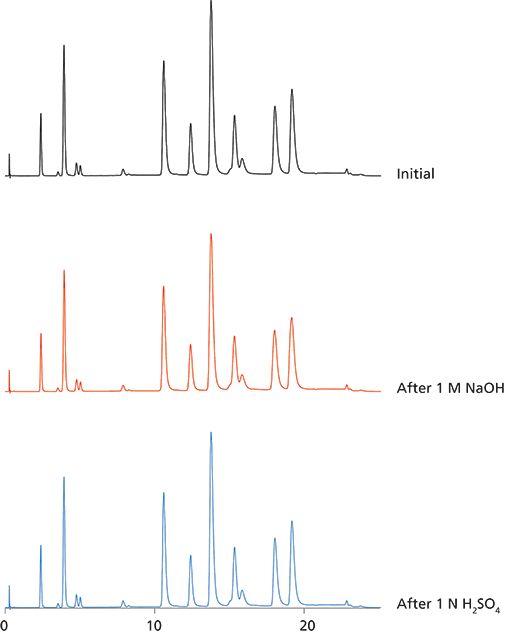
Figure 1: Separation of standard mix after 200 column volumes 1 M NaOH, 1 M H2SO4.
Conclusion
Mobile phase pH is a useful tool in methods development, particularly for separation of neutral forms of amines or other organic bases under alkaline conditions. Although a few more recent C18 columns boast stability in alkaline pH, column life is still considerably shorter than if used under more favorable conditions. On the other hand, the PRP-C18 has genuine pH and chemical stability. The stationary phase is devoid of free silanols, does not strip, bleed, or dissolve at any pH, and therefore can be expected to perform reliably and reproducibly throughout the extended life of the column, regardless of mobile phase conditions.

Hamilton Company
4970 Energy Way, Reno, NV 89502
tel. (800) 648-5950, fax (775) 858-3026
Website: www.hamiltoncompany.com

Analysis of Greenhouse Gases by Gas Chromatography
May 15th 2024This application note demonstrates the use of SCION's 8500GC system for the analysis of key greenhouse gases—carbon dioxide, methane, and nitrous oxide—in a single atmospheric air matrix. Highlighting the system's excellent sensitivity and repeatability, this method is crucial for understanding emission sources and combating climate change.
Analysis of DEHP in Drinking Water by HPLC-DAD
May 15th 2024This application note outlines a method for detecting low levels of 1,4-Dioxane, a likely carcinogenic compound, in drinking water using SCION GC-MS technology. It details the procedure for analyzing water samples spiked with 1,4-Dioxane, achieving excellent sensitivity and low detection limits, demonstrating the method's effectiveness for environmental monitoring.
Plant Protection Product Impurity Screening by GC-FID with GC-MS Confirmation
May 15th 2024This application note from SCION Instruments delves into the meticulous screening of impurities in plant protection products using GC-FID, with GC-MS for confirmation. It emphasizes the necessity of identifying and quantifying impurities to comply with regulatory standards in industries such as pharmaceuticals, food, and agriculture. The study specifically examines eugenol, showcasing how to determine significant impurities for regulatory submission. The approach integrates GC-MS and analytical standards, ensuring precise impurity identification and quantification critical for product approval.