Separation of a Mix of Acidic, Basic, and Neutral Compounds at High pH Conditions
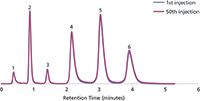
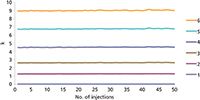
The unique surface chemistry of the Flare diamond core-shell column combines ionic and hydrophobic separation mechanisms to effectively retain a variety of chemical species in a single run.
The unique surface chemistry of the Flare diamond core-shell column combines ionic and hydrophobic separation mechanisms to effectively retain a variety of chemical species in a single run.

HPLC Conditions
Column Name: Flare C18 Mixed-Mode Column
Dimensions: 4.6 × 33 mm, 3.6 µm, 180 Å
HPLC System: Waters® 1525 Binary Pump
Injection Volume: 5 µL
Detection: UV at 254 nm
Flow Rate: 1.0 mL/min
Separation Mode: Isocratic
Mobile Phase: A: 10 mM phosphate buffer, pH 12; B: acetonitrile A/B (70:30)
Temperature: 35 °C
Analytes:
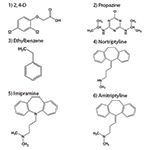
Conclusions
1) Fast separation at high pH
2) Separation possible with 100% aqueous mobile phase
3) Straightforward method transfer to any system

Diamond Analytics
11260 South 1600 West, Orem, UT 84058
tel. (801) 235-9001, fax (801) 235-9141
Website: diamond-analytics.com

Analysis of Greenhouse Gases by Gas Chromatography
May 15th 2024This application note demonstrates the use of SCION's 8500GC system for the analysis of key greenhouse gases—carbon dioxide, methane, and nitrous oxide—in a single atmospheric air matrix. Highlighting the system's excellent sensitivity and repeatability, this method is crucial for understanding emission sources and combating climate change.
Analysis of DEHP in Drinking Water by HPLC-DAD
May 15th 2024This application note outlines a method for detecting low levels of 1,4-Dioxane, a likely carcinogenic compound, in drinking water using SCION GC-MS technology. It details the procedure for analyzing water samples spiked with 1,4-Dioxane, achieving excellent sensitivity and low detection limits, demonstrating the method's effectiveness for environmental monitoring.
Plant Protection Product Impurity Screening by GC-FID with GC-MS Confirmation
May 15th 2024This application note from SCION Instruments delves into the meticulous screening of impurities in plant protection products using GC-FID, with GC-MS for confirmation. It emphasizes the necessity of identifying and quantifying impurities to comply with regulatory standards in industries such as pharmaceuticals, food, and agriculture. The study specifically examines eugenol, showcasing how to determine significant impurities for regulatory submission. The approach integrates GC-MS and analytical standards, ensuring precise impurity identification and quantification critical for product approval.