Analysis of "Broad Spectrum" UVA and UVB Components in Sun Care Products for Compliance with New FDA Regulations
The Application Notebook
The FDA has made changes to how products containing sunscreen are labeled in the U.S. to ensure they meet the new regulations set forth for safety and effectiveness. The new regulations will require companies that want to use the "broad spectrum" label to test for both UVA and UVB protection.
The FDA has made changes to how products containing sunscreen are labeled in the U.S. to ensure they meet the new regulations set forth for safety and effectiveness. The new regulations will require companies that want to use the "broad spectrum" label to test for both UVA and UVB protection. Previous regulations only dealt with preventing sunburn which is primarily due to UVB radiation but did not address UVA which protects against early aging and skin cancer. All products that claim to provide broad spectrum SPF protection are regulated as sunscreen products. Therefore, the regulations the FDA has developed for over the counter (OTC) sunscreen products apply to cosmetics, moisturizers, lip balms, and shampoos labeled with SPF values.

Table I: Experimental Conditions
Results
Conclusion
The methodology in Figure 1 provides one test method to determine both UVA and UVB compounds most commonly used in the sun care industry. Figure 2 is a sun care spray run with the same analytical conditions eluting six UVA/B compounds. All UVB and UVA compounds are eluted in a single run and can be quantitated in the ranges recommended by the FDA (SPF 15–50+). Only broad spectrum products with an SPF value of 15 or higher can claim to reduce the risk of skin cancer and early skin aging if used as directed with other sun protection measures.
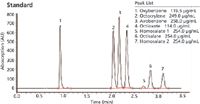
Figure 1: All UVA and UVB compounds are eluted in a single run using a Brownlee SPP column with an optimized PerkinElmer HPLC system
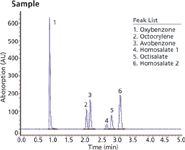
Figure 2: SPF 50 sun care spray shows elution of both UVA/B compounds.
References
(1) U.S. Food and Drug Administration, 2012. Information for Consumers, 2012. Retrieved from http://www.fda.gov/Drugs/ResourcesForYou/Consumers/BuyingUsingMedicineSafely/UnderstandingOver-the-CounterMedicines/ucm258468.htm.
PerkinElmer
940 Winter Street, Waltham, MA 02451
Tel. (800) 762-4000, fax (203) 944-4904
Website: www.perkinelmer.com

Analysis of Greenhouse Gases by Gas Chromatography
May 15th 2024This application note demonstrates the use of SCION's 8500GC system for the analysis of key greenhouse gases—carbon dioxide, methane, and nitrous oxide—in a single atmospheric air matrix. Highlighting the system's excellent sensitivity and repeatability, this method is crucial for understanding emission sources and combating climate change.
Analysis of DEHP in Drinking Water by HPLC-DAD
May 15th 2024This application note outlines a method for detecting low levels of 1,4-Dioxane, a likely carcinogenic compound, in drinking water using SCION GC-MS technology. It details the procedure for analyzing water samples spiked with 1,4-Dioxane, achieving excellent sensitivity and low detection limits, demonstrating the method's effectiveness for environmental monitoring.
Plant Protection Product Impurity Screening by GC-FID with GC-MS Confirmation
May 15th 2024This application note from SCION Instruments delves into the meticulous screening of impurities in plant protection products using GC-FID, with GC-MS for confirmation. It emphasizes the necessity of identifying and quantifying impurities to comply with regulatory standards in industries such as pharmaceuticals, food, and agriculture. The study specifically examines eugenol, showcasing how to determine significant impurities for regulatory submission. The approach integrates GC-MS and analytical standards, ensuring precise impurity identification and quantification critical for product approval.