Special Issues
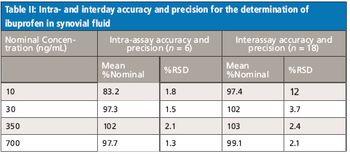
Published methods for the determination of ibuprofen in biological fluids by liquid chromatography (LC)–UV or LC–mass spectrometry (MS)-MS have quantitation ranges consistent with the relatively high but typical ibuprofen dose (200–800 mg), generally having lower limits of quantitation in the low micrograms-per-milliliter range. For the analysis of plasma and synovial fluid samples from preclinical (miniature swine) studies utilizing a novel ibuprofen dosage form, LC–MS-MS methods were developed and validated over the 10–1000 ng/mL range. Ibuprofen undergoes biotransformation to ibuprofen acyl glucuronide and sublimes under routine bioanalytical sample handling conditions. Procedures were implemented to minimize the impact of these potential liabilities.

