Biopharmaceuticals and Protein Analysis
Latest News
Latest Videos

Shorts

More News

Torgny Fornstedt, Jörgen Samuelsson, and Martin Enmark discuss a novel machine learning workflow for oligonucleotide analysis that is helping to enhance method development and impurity profiling efficiency.

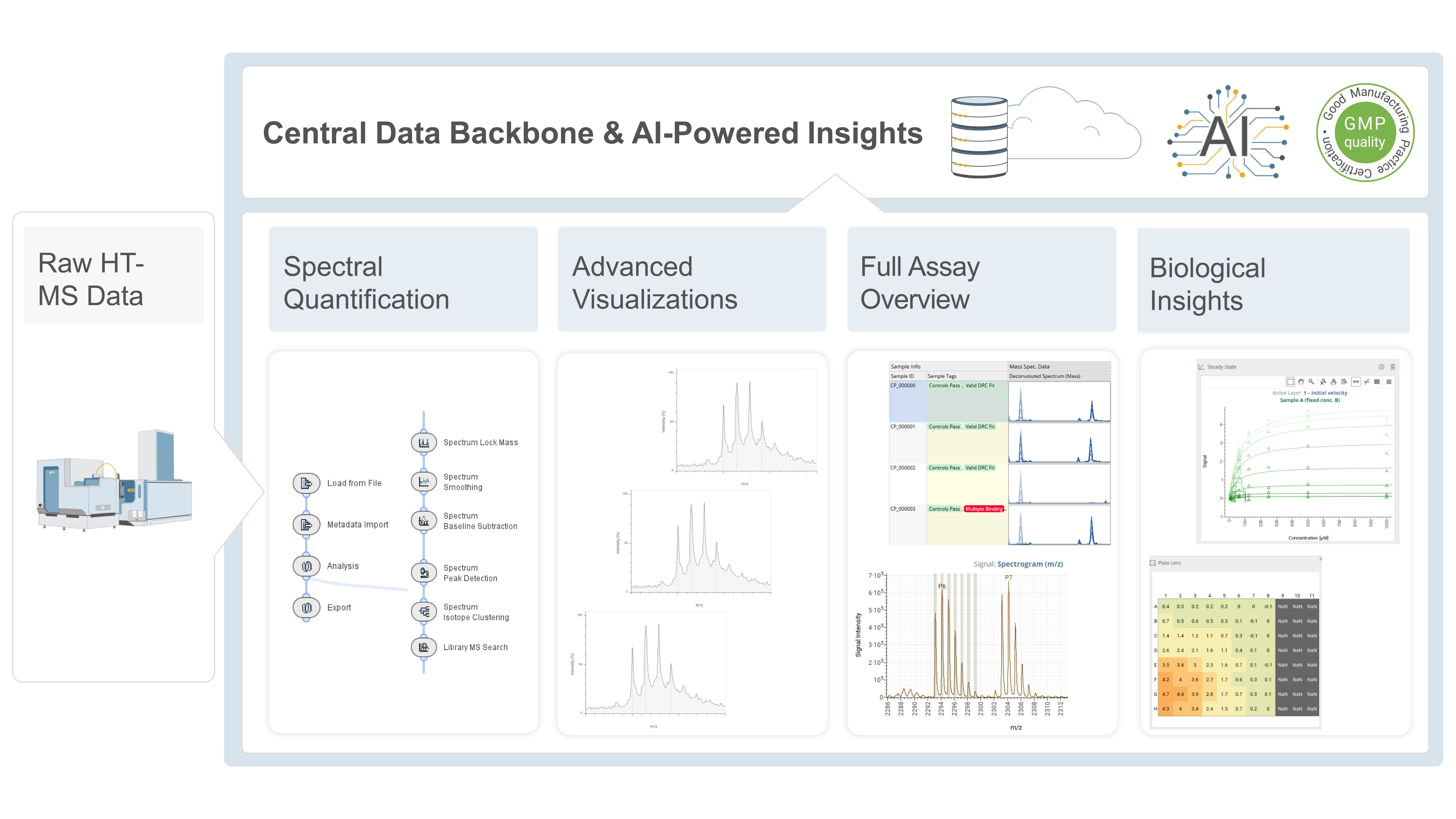
Analyzing Small RNA-based Therapeutics and Their Process Impurities Using Fast and Sensitive LC–HRMS
Discover innovative LC–HRMS methods for analyzing RNA-based therapeutics, enhancing biopharma development and regulatory compliance in drug characterization.
Static and dynamic electrostatic repulsion-reversed phase (ERRP) has been compared for the separation of peptide impurities.

The advancements in HILIC–MS for oligonucleotide analysis are discussed, highlighting its potential as a viable alternative to traditional methods.

A reversed-phase liquid chromatography–charged aerosol detector (RPLC–CAD) method was developed and validated per ICH Q2(R1) guidelines for simethicone quantification.
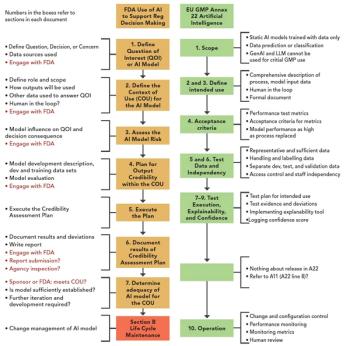
Does AI/ML help to enhance chromatographic peak integration? What do regulators say? Are you being seduced by technology?

SFC Europe 2025—the 19th International Conference on Packed Column Supercritical Fluid Chromatography and Related Techniques—will be held in Basel, Switzerland, on October 5–7, 2025. LCGC International spoke to John Reilly, Maria Kristina Parr, and Larry Miller about the important role that SFC now plays in life science analysis
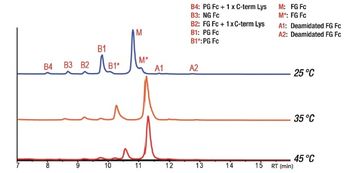
This article discusses the advantages of a native anion exchange method coupled to mass spectrometry (MS) for charge heterogeneity analysis of immunoglobulin G4 (IgG4)-based mAbs, which are currently being studied more intensively.

This article explores advanced oligonucleotide analysis techniques, including chromatography methods for therapeutic RNA, siRNA, and mRNA, enhancing nucleic acid research.
Explore the complexities of bispecific antibodies and multispecific biologics, focusing on production, purification, and analytical challenges in biopharmaceuticals.

In this study, we applied a high-sensitivity MAPPs workflow using magnetic bead-based HLA-DR/DP/DQ immunoprecipitation and high-resolution mass spectrometry (HRMS) to assess the immunogenicity risk of six marketed mAbs across a panel of 10 HLA-typed donors.

This article reviews literature related to 2D-LC applications in the biopharmaceutical field over the past six years (2018-2024).

Curcumin is a polyphenol believed to have anticancer effects. To better quantify its existence in plasma, reversed-phase high-performance liquid chromatography (RP-HPLC) was used to create a new analytical approach for quantifying this substance.

In this article, you’ll find some of the top content that was published on LCGC this week, including calls for new chromatography technologies and interviews with rising stars in the field.

To create a new approach for monitoring and quantifying free fatty acids (FFAs), Chinese researchers combined stable isotope derivatization coupled with liquid chromatography–triple quadrupole mass spectrometry (ID-LC-QQQ-MS).

Enantiomer self-disproportionation can occur during various scientific processes. To understand temperature’s relationship with this phenomenon, researchers studied how temperature affected SDE in chromatographic separation.

Researchers from Comenius University Bratislava used LC–MS/MS to analyze albendazole substances in plasma samples.

Researchers at the Institute of Marine Research in Bergen, Norway used liquid chromatography–mass spectrometry to analyze resolvins in fish and human cells.

A new approach for purifying immunoglobulin-based therapies was created by Pfizer researchers.

To detect popular party drugs in human waste, researchers combined headspace solid-phase microextraction and solid-phase microextraction to detect these substances and their metabolites.
University of Geneva researchers led a collaboration for testing different size-exclusion chromatography columns to compare the capability for characterizing mRNA.

Researchers at LUT University highlight challenges in calculating adsorption values during aqueous chromatography and explore new methods to close this knowledge gap.

To better analyze polyphenols, scientists from Sapienza University of Rome examined the capabilities of supercritical fluid chromatography (SFC), comparing it to traditional methods.

Countercurrent chromatography systems were tested and combined to optimize their capacity for handling non-polar and polar substances.

A new study done by scientists in Switzerland and Sweden investigated the use of automated systems to develop multispecific antibodies.