
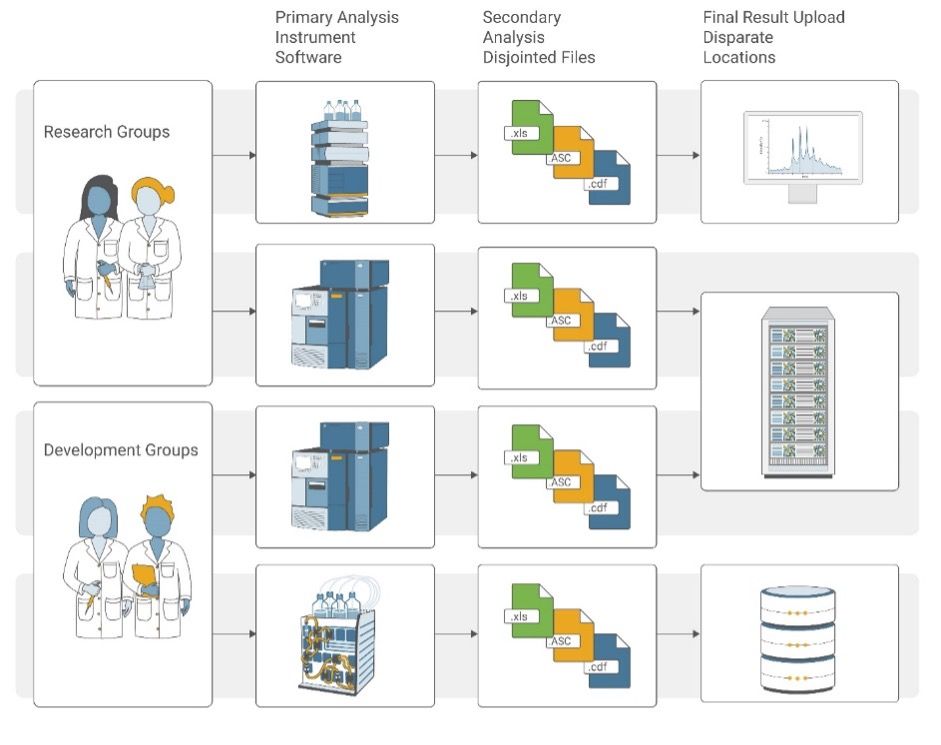
Data Acquisition, Handling, and Archiving
Latest News
Latest Videos

More News

The 11th International Symposium on Recent Advances in Food Analysis will be in Prague, Czech Republic from 5–8 November 2024.

In preparation for ASMS 2024, we recently interviewed Uri Keshet of the University of California, Davis about his upcoming presentation and expectations for the conference.

In this edition of The LCGC Blog, Katelynn Perrault and James Harynuk discuss the technological needs one faces when setting up a workstation for chromatography data analysis.

This article examinations the determination of retention time and why it's important.

Tired of life as your analytical results are always out of specification (OOS)? Fed up with yet another laboratory investigation? Get those rotten chromatograms to generate passing results by learning ways to manipulate peak integration from the experts... and now I have your undivided attention...and how reviewers, QA and inspectors can detect them!

Are you interested in ChatGPT's vast potential? Or are you getting tired of reading or hearing about ChatGPT?

Webinar Date/Time: Mon, Oct 23, 2023 11:00 AM EDT

Are you interested by ChatGPT's vast potential? Or are you getting tired of reading or hearing about ChatGPT? If the latter, this blog post might give you some tips and tricks on taking advantage of ChatGPT while avoiding its application beyond its intended scope.

This blog addresses the essential role software has in shaping the present and future of chromatography for the better.

A new publication introduces a groundbreaking automated system for high-throughput mass spectrometry analysis, reshaping the future of biotherapeutic characterization.

We interviewed an AI program (ChatGPT) for LCGC North America asking questions about AI and its role in various applications for separation science to include data analysis, and high performance liquid chromatography (HPLC), hydrophilic-interaction chromatography (HILIC), reversed-phase liquid chromatography (RPLC), liquid chromatography–mass spectrometry (LC–MS), gas chromatography–mass spectrometry (GC–MS), high resolution mass spectrometry (HRIM–MS), high resolution tandem mass spectrometry (HRMS/MS), and related topics.

Separation science will be at the core of the artificial intelligence (AI) revolution in science.

Determining a theoretical number of plates is an effective tool to ensure that the column is still applicable for the desired separation.

Webinar Date/Time: Thu, Apr 6, 2023 11:00 AM EDT

Webinar Date/Time: Day One, Monday, March 13, 2023 Day Two, Tuesday, March 14, 2023 Day 1: 10:00 am – 4:30 pm EST Day 2: 10:00 am – 4:30 pm EST

The scope of data integrity is shown by the data integrity model, which sits within the overall pharmaceutical quality system of a regulated laboratory.

A snapshot of key trends and developments in data handling according to selected panellists from the chromatography sector.
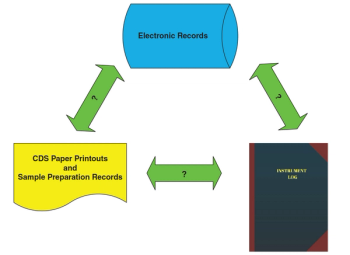
The term orphan data is used frequently in the context of data integrity. What does it mean for chromatography data systems? How can we prevent or detect orphan data?

What do the draft publications ICH Q2(R2) and Q14 for analytical procedure validation and development mean for a regulated GMP laboratory?

Our annual review of new high performance liquid chromatography instruments, mass spectrometry systems, and data processing software.

A balance printout is a fixed record, and is also called static data. But how static are static data when the weight is used in a chromatographic analysis? Also, have some regulatory data integrity guidance documents failed to comply with their own regulations?

Are we ready to let software and instruments take over the art of separations?

In September, the World Health Organization (WHO) issued a new guidance document on Good Chromatography Practices. What guidance does it contain and is it useful? Has the document failed its system suitability test (SST) acceptance criteria?





