LCGC North America
What are the characteristics and relevant considerations for the development and validation of a stability-indicating method?

LCGC North America
What are the characteristics and relevant considerations for the development and validation of a stability-indicating method?

LCGC Europe
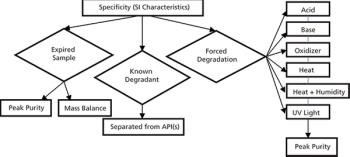
In the pharmaceutical industry, the demonstration of drug substance (DS) or drug product (DP) stability over the shelf life is an important aspect of the regulatory requirements.
![Roy figure 5-[41827930]-{585410}-746304-1416911904998.gif](https://cdn.sanity.io/images/0vv8moc6/chroma/5e8a7a8b42be4c1287e42a9e19c61a898804cfe5-700x592.gif?w=350&fit=crop&auto=format)
Special Issues
A discussion of active pharmaceutical ingredient (API) selection, drug product development, and mass spectrometry instrumentation