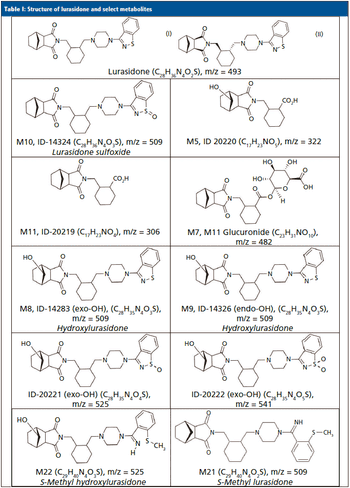
For lurasidone treatment adherence testing, an untargeted high-resolution mass spectrometry method was employed, using known positive human urine samples to identify the lurasidone metabolites and their relative abundance in urine.

For lurasidone treatment adherence testing, an untargeted high-resolution mass spectrometry method was employed, using known positive human urine samples to identify the lurasidone metabolites and their relative abundance in urine.

An investigation of C18 and phenyl-hexyl column chemistries for definitive identification of 13 synthetic cannabinoid metabolites in patient samples.
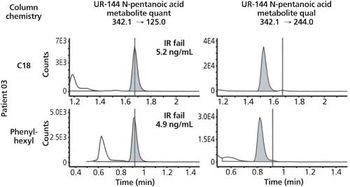
In recent years, synthetic cannabinoids (or “spice”) have experienced a boom in popularity. The negative health effects of these drugs coupled with their increasing popularity led to placement onto Schedule I by the DEA. In response, the chemists behind these illicit compounds frequently invent new compounds to circumvent the law. Thus, new classes and new examples within classes of “spice” continue to become available for illicit use. In this paper, we examine the use of two different column chemistries (C18 and phenyl-hexyl) in an effort to definitively identify synthetic cannabinoid compounds in patient samples.
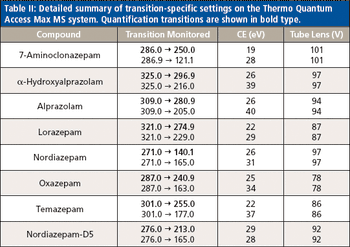
A new method was developed and validated using automated on-line solid-phase extraction (SPE) with tandem mass spectrometry (MS). Urine samples were enzyme-hydrolyzed and diluted before detection. The validated method was applied to positive authentic urine samples to evaluate concordance with high performance liquid chromatography (HPLC)–MS-MS results.
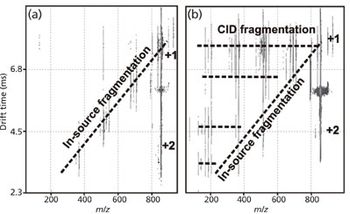
Ion mobility–mass spectrometry (IM-MS) is outlined as a separations method, several examples of the utility of IM-MS for complex biological measurements are illustrated, and the implications of this approach for systems biology research are discussed.

Published: July 1st 2011 | Updated:
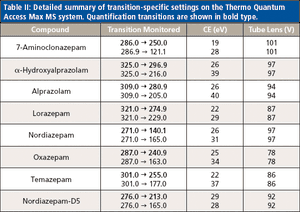
Published: May 1st 2015 | Updated:
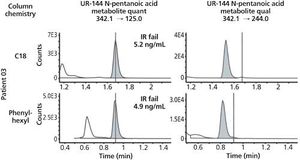
Published: April 1st 2016 | Updated:
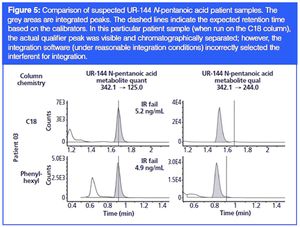
Published: May 18th 2016 | Updated:
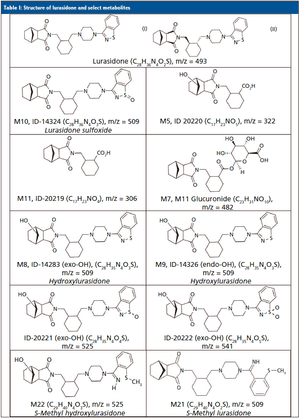
Published: May 1st 2019 | Updated: