
Biomimetic HPLC retention data can be used to measure how a compound will bind to proteins and phospholipids in vivo.

Biomimetic HPLC retention data can be used to measure how a compound will bind to proteins and phospholipids in vivo.
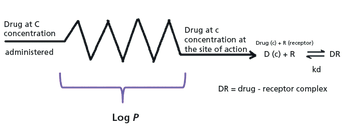
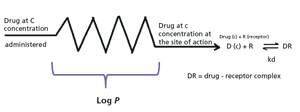
Drug discovery can be accelerated by chromatographic profiling of analogs by measuring their nonspecific binding to proteins and lipids.
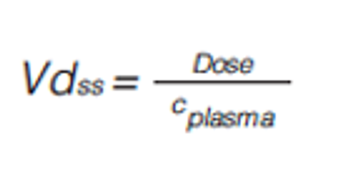
The biomimetic gradient retention time measurements on C18, immobilized artificial membrane (IAM), human serum albumin (HSA), and acid-glycoprotein (AGP) stationary phases can be used to characterize compounds partitioning into phospholipids and proteins. The data obtained can then be used in equations to estimate the in vivo plasmaÐtissue distribution of the compounds measured. The plasma protein binding, brain tissue binding, and in vivo drug efficiency can also be calculated using the biomimetic chromatographic data.

The drug discovery process can be accelerated by chromatographic profiling of analogs by measuring their nonspecific binding to proteins and lipids and then by modelling in vivo distribution. A balanced potency and chromatographically determined membrane and protein binding ensure the selection of compounds with the highest probability to show the desired in vivo distribution behaviour for efficacy and reduced toxicity. The first part of the article will discuss the high performance liquid chromatography (HPLC)-based measurements of lipophilicity and biomimetic properties, while the second part will discuss the models derived from the measured data of known drug molecules and drug discovery compounds.

Fast, reliable measurements of lipophilicity has a great impact on the compounds' selection process at an early stage of dug discovery. The guest columnit examines HPLC methods for this purpose.

Published: March 1st 2007 | Updated:
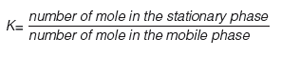
Published: February 1st 2018 | Updated:

Published: May 1st 2018 | Updated:
Published: June 1st 2018 | Updated:

Published: August 1st 2018 | Updated: