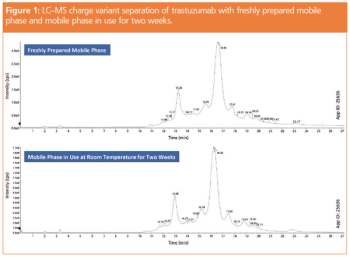
The utility of native high-resolution mass spectrometry (HRMS) in intact protein characterization is rapidly growing because of advances in both ion-exchange chromatography (IEC) as well as MS-compatible buffer systems. MS is a critical component of biotherapeutic characterization, but its combination with traditional chromatographic separations, such as size-exclusion chromatography (SEC) and IEC, has been slow because of the predominant use of high salt mobile phases, which are incompatible with MS. Recently reported methods using cation-exchange chromatography (CEX) with volatile buffer systems for pH gradient elution has given researchers the ability to use these chromatographic techniques with MS detection. In this article a robust, MS-compatible buffer system for high sensitivity IEC with pH gradient elution for charge variant analysis of intact monoclonal antibodies (mAbs) is described.





