The benefits of a robustness assessment for the analysis of a NIST mAb using a wide pore C4 LC column are described.

The benefits of a robustness assessment for the analysis of a NIST mAb using a wide pore C4 LC column are described.
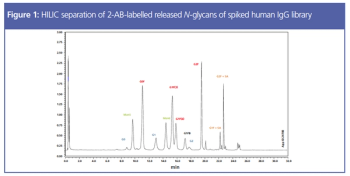
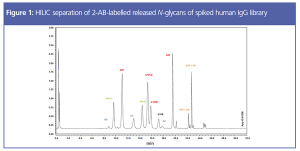
Post-translational modifications are potential critical quality attributes (pCQAs) routinely assessed in biotherapeutic development. Glycosylation is one of the most important attributes to assess because it affects protein function as well as antigen receptor binding. N-glycosylation of asparagine residues is the most common pCQA assessed during monoclonal antibody (mAb) therapeutic development. There are a few protocols to assess and quantitate N-glycans, but the most common approach is through an enzymatic release and labelling procedure, followed by separation and detection. This article demonstrates the method development considerations for sample preparation and chromatographic analysis of N-glycans of therapeutic mAbs.

This article describes the importance of magnetic bead and chromatographic media selection for hybrid LBA/LC–MS/MS workflows.
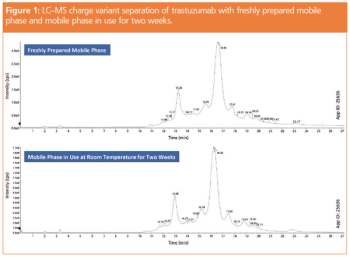
The utility of native high-resolution mass spectrometry (HRMS) in intact protein characterization is rapidly growing because of advances in both ion-exchange chromatography (IEC) as well as MS-compatible buffer systems. MS is a critical component of biotherapeutic characterization, but its combination with traditional chromatographic separations, such as size-exclusion chromatography (SEC) and IEC, has been slow because of the predominant use of high salt mobile phases, which are incompatible with MS. Recently reported methods using cation-exchange chromatography (CEX) with volatile buffer systems for pH gradient elution has given researchers the ability to use these chromatographic techniques with MS detection. In this article a robust, MS-compatible buffer system for high sensitivity IEC with pH gradient elution for charge variant analysis of intact monoclonal antibodies (mAbs) is described.

Within the broad scope of analytical techniques required to characterize a protein, chromatographic methods have shifted towards high-flow analyses that can drop development time significantly. However, fast analytical methods for charge heterogeneity have lagged in development because current column technologies are ultrahigh-pressure liquid chromatography (UHPLC)-incompatible. This article will demonstrate the development of a high-flow method for charge variant analysis made possible through a bioinert titanium column flow path.

Published: November 12th 2018 | Updated:

Published: November 6th 2019 | Updated:

Published: February 5th 2020 | Updated:
Published: May 6th 2020 | Updated: