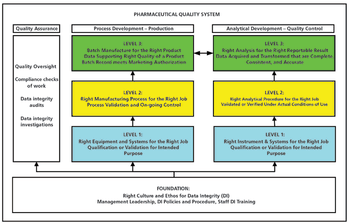
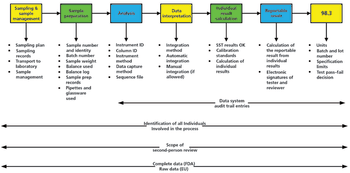
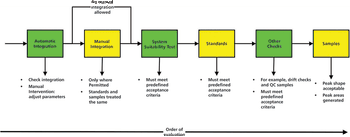
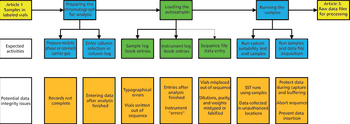
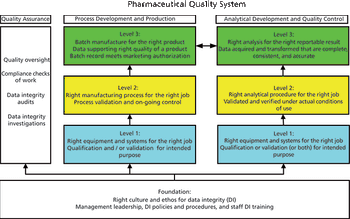
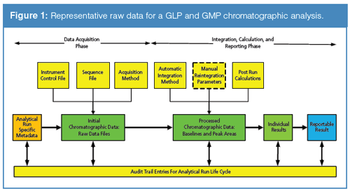
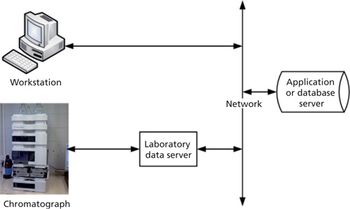
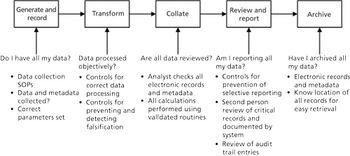
This is the final part of a trilogy looking at the impact of the new USP on analytical instrument qualification (AIQ) on data integrity in a regulated chromatography laboratory. This part will focus on the performance qualification (PQ) portion of the 4Qs model and, specifically, monitoring and requalification of chromatographs.