
A review of FAST’s virtual e-lecture series and discussions.

A review of FAST’s virtual e-lecture series and discussions.

Clinical metabolomics requires analytical methods that provide both high resolution and high throughput. In this review, we assess the promise of a variety of techniques-including HILIC, SFC, multidimensional LC, ion-mobility mass spectrometry, and data-independent acquisition mass spectrometry methods-to meet those needs.
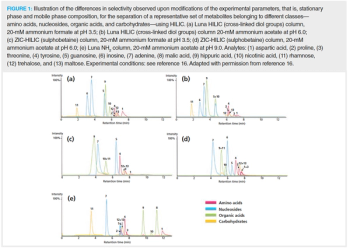
This review article discusses the novel separation and detection strategies that are considered promising in clinical metabolomics to enhance the metabolome coverage. It includes hydrophilic interaction chromatography (HILIC), supercritical fluid chromatography (SFC), multidimensional LC approaches, as well as ion-mobility mass spectrometry (IM-MS) and data-independent acquisition (DIA) analysis methods.

CZE–ESI‑TOF‑MS for the characterization of the mAb infliximab and its variants is presented. Infliximab was analyzed using a middle-up approach involving either reduction or digestion with the enzyme IdeS. A multilayer capillary coating of PB-DS‑PB in combination with a background electrolyte of 40% acetic acid provided efficient separation of the obtained antibody fragments, that is, LC and HC, as well as F(ab’)2 and Fc/2 parts. C-terminal lysine variants were also resolved. Recorded mass spectra of HC and Fc/2 fragments permitted assignment of 13 glycoforms and provided a quantitative profile, with G0F the most abundant glycoform (~50%). CZE–ESI-TOF-MS represents an efficient means for the straightforward analysis of a monoclonal antibody and its proteoforms.

This information is supplementary to the article “Middle-Up Characterization of the Monoclonal Antibody Infliximab by Capillary Zone Electrophoresis-Mass Spectrometry” that was published in the March 2019 issue of LCGC Europe.

An overview of the important and versatile role that capillary electrophoresis coupled to mass spectrometry plays in the biopharmaceutical analysis.

An overview of the important and versatile role that capillary electrophoresis coupled to mass spectrometry plays n biopharmaceutical analysis.

CE–MS using noncovalent coated capillaries provides advantages for purity and stability analysis of biopharmaceuticals.

Published: May 6th 2021 | Updated:

Published: November 2nd 2012 | Updated:

Published: October 1st 2012 | Updated:

Published: June 1st 2012 | Updated:

Published: February 28th 2019 | Updated:

Published: March 1st 2019 | Updated: