Articles by Teri Dattilio

The United States Food and Drug Administration (U.S. FDA) and the European Union (EU) have published regulations and provided guidance on the use of chemicals in food packaging materials. Tekmar provides method parameters for the HT3â„¢ Automated Headspace Analyzer for the determination of VOCs that may be contained in food contact packaging materials under various conditions. Temperature and mixing parameters were investigated to determine which VOCs may be released when subjected to these conditions.
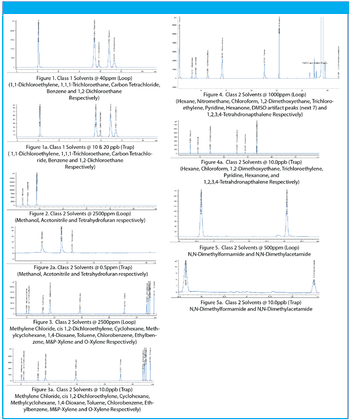
Residual solvents in pharmaceuticals are defined as volatile organic chemicals that are used or produced in the manufacture of drug substances, excipients or in the preparation of drug products. Because residual solvents do not provide therapeutic benefits, they should be removed, to the extent possible. Drug products should contain no higher levels of residual solvents than can be supported by safety data. Looking forward to the implementation of a revised USP <467> method, Teledyne Tekmar evaluated the new protocol, therefore this application will comply with the procedure and criteria changes set forth in the USP30 NF25, Second Supplement (effective December 1, 2007) and the interim revision announcement. (1)




