
Tore Vehus
Articles by Tore Vehus

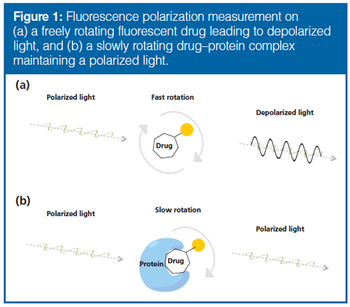
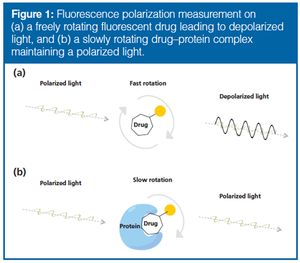
Fluorescence polarization (FP) is a highly regarded technique for studying drug–protein interactions, but has limited value regarding protein mixtures. As a novel approach to drug target discovery, the possibility of combining FP with liquid chromatography (LC) was explored. Nondenaturing protein LC principles such as size-exclusion chromatography (SEC), hydrophobic interaction chromatography (HIC), and ion exchange chromatography (IEX) were found to be orthogonal and compatible with FP because the mobile phases used do not negatively affect detection. For simple protein mixtures, the SEC/HIC/IEX–FP approach was able to identify tankyrase as the target of a triazole-based inhibitor of the Wnt signaling pathway, which is heavily associated with colon cancer. However, the total peak capacity of the three LC dimensions was not sufficient to resolve at cell-proteome level, calling for higher resolution of intact proteins to enable stand-alone drug target discovery with LC and FP.
Latest Updated Articles
 Supplementary Information: Combining HIC, SEC, and IEX with Fluorescence Polarization for Drug Target Discovery
Supplementary Information: Combining HIC, SEC, and IEX with Fluorescence Polarization for Drug Target DiscoveryPublished: May 5th 2017 | Updated:
 Combining HIC, SEC, and IEX with Fluorescence Polarization for Drug Target Discovery
Combining HIC, SEC, and IEX with Fluorescence Polarization for Drug Target DiscoveryPublished: May 1st 2017 | Updated:
