Articles by Vishwanath Hebbi

In this article, we discuss the use of CE-MS (sheath flow interface) for analysis of intact proteins as well as of protein digests. We discuss the unique aspects that the user needs to be aware of while testing biotherapeutics versus small molecule drugs. We also highlight that the optimization of CE and MS parameters together result in the creation of a more robust and reproducible protein analysis approach. Finally, we list some of the most common errors that are likely to occur during CE-MS analysis and suggest ways to overcome them.
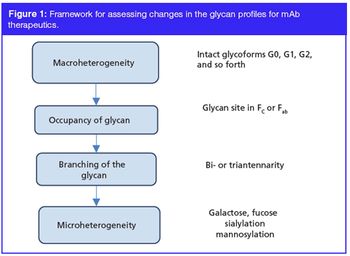
Glycosylation of monoclonal antibody (mAb) therapeutics is widely recognized by the regulators and the industry as a critical quality attribute (CQA). Hence, it is necessary that glycosylation is measured and adequately controlled during production. This instalment reviews the various process parameters and raw material attributes that affect glycosylation, as well as the different analytical tools that are used for characterization, with greater emphasis on the chromatographic methods of analysis. Key recent advancements that have occurred in the past five years are also discussed briefly. While significant progress has been made in the monitoring of glycosylation, its real time control has yet to be demonstrated.

Glycosylation of monoclonal antibody (mAb) therapeutics is widely recognized by the regulators and the industry as a critical quality attribute (CQA). Hence, it is necessary that glycosylation is measured and adequately controlled during production. This installment reviews the various process parameters and raw material attributes that affect glycosylation, as well as the different analytical tools that are used for characterization, with greater emphasis on the chromatographic methods of analysis. Key recent advancements that have occurred in the past five years are also discussed briefly. While significant progress has been made in the monitoring of glycosylation, its real time control has yet to be demonstrated.






