
LCGC North America
Multidimensional separations, in which two or more separation methods are coupled, are a valuable analytical tool for higher peak capacity and improved selectivity for the analysis of complex samples like biotherapeutics.

LCGC North America
Multidimensional separations, in which two or more separation methods are coupled, are a valuable analytical tool for higher peak capacity and improved selectivity for the analysis of complex samples like biotherapeutics.

LCGC North America
Advanced separation and mass spectrometry methods enable comprehensive profiling of the inherent glycan heterogeneities of protein therapeutics. In particular, reversed-phase HPLC–based multiattribute methods (MAMs) provide a wealth of information, and other techniques, such as HILIC and CE-MS, also continue to evolve.
LCGC North America
Coupling CE with MS presents some challenges. Here, we discuss the advantages of CE–MS over LC–MS, and the parameters that are important to obtain a stable CE–MS profile.

LCGC North America
Determining the higher order structure of a protein pharmaceutical is important. Here, we review the approaches for HOS determination that are currently receiving the most attention in the literature and at scientific meetings.
LCGC North America
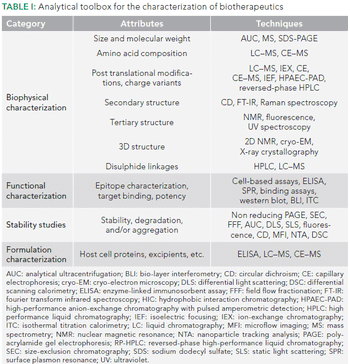
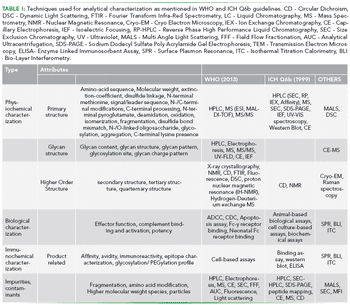
The analytical techniques used for characterizing biotherapeutics have evolved. We review the utility of the traditional tools and discuss the new, orthogonal techniques that are increasingly being used.

LCGC North America
To ensure the reliable and accurate characterization of biotherapeutics, an arsenal of orthogonal analytical techniques is needed.

LCGC North America
A look at techniques for charge-variant analysis of monoclonal antibodies and the question of whether pH gradients are really better than salt gradients

LCGC North America
Several key applications of biolayer interferometry in pharmaceutical development have emerged recently. Here, we evaluate its use for easuring product titer from fermentation, and compare the strengths and weaknesses of the technique to those of HPLC.

LCGC Asia Pacific
All agencies have issued varying guidances for the approval of recombinant biosimilars of biopharmaceuticals, and all submittals are considered on a case-by-case basis. This instalment of “Focus on Biopharmaceutical Analysis” looks at the best methodologies for demonstrating their analytical comparability.

LCGC North America
All agencies have issued varying guidances for the approval of recombinant, biosimilars of biopharmaceuticals. However, their impact or meaning is in our understanding and that all submittals are considered on a case-by-case basis.

LCGC North America
In this article, we discuss the use of CE-MS (sheath flow interface) for analysis of intact proteins as well as of protein digests. We discuss the unique aspects that the user needs to be aware of while testing biotherapeutics versus small molecule drugs. We also highlight that the optimization of CE and MS parameters together result in the creation of a more robust and reproducible protein analysis approach. Finally, we list some of the most common errors that are likely to occur during CE-MS analysis and suggest ways to overcome them.

The Column
A review of the 10th Balaton Symposium on High-Performance Separation Methods, which was held 2–4 September 2015 at the Hotel Azúr, Siófok, Hungary.

LCGC North America
Glycosylation of monoclonal antibody (mAb) therapeutics is widely recognized by the regulators and the industry as a critical quality attribute (CQA). Hence, it is necessary that glycosylation is measured and adequately controlled during production. This installment reviews the various process parameters and raw material attributes that affect glycosylation, as well as the different analytical tools that are used for characterization, with greater emphasis on the chromatographic methods of analysis. Key recent advancements that have occurred in the past five years are also discussed briefly. While significant progress has been made in the monitoring of glycosylation, its real time control has yet to be demonstrated.

LCGC North America
A systematic approach for formation of sigmoidal gradient for analysis of biopharmaceutical proteins has been reviewed and discussed together with two case studies. The first application involves HPLC separation of the various product related variants of a microbial expressed biotherapeutic, granulocyte colony stimulating factor (GCSF). The second case study involves separation of the charge heterogeneity related variants for a monoclonal antibody (mAb) biotherapeutic product. In all cases, it is observed that the use of sigmoidal gradient successfully reduces the analysis time significantly (from 70 minutes to 15 minutes for GCSF and from 40 minutes to 4 minutes for the mAb) while retaining the selectivity and the resolution.

LCGC North America
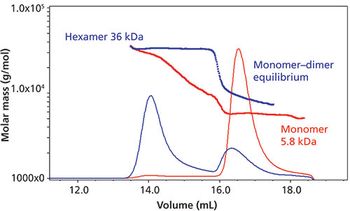
Here in part II of our series on assessing protein aggregation, we provide an overview of best practices for achieving this goal, including the importance of using a multimethod approach.Here in part II of our series on assessing protein aggregation, we provide an overview of best practices for achieving this goal, including the importance of using a multimethod approach.
LCGC Europe
Various analytical approaches and instrumentation for low?molecular-weight (MW), noncovalent, aggregates of proteins are discussed.

LCGC North America
Most analytical approaches used for the determination of low-molecular-weight noncovalent aggregates in protein pharmaceuticals suffer from important limitations. This first part of this two-part column series discusses those limitations.

LCGC North America
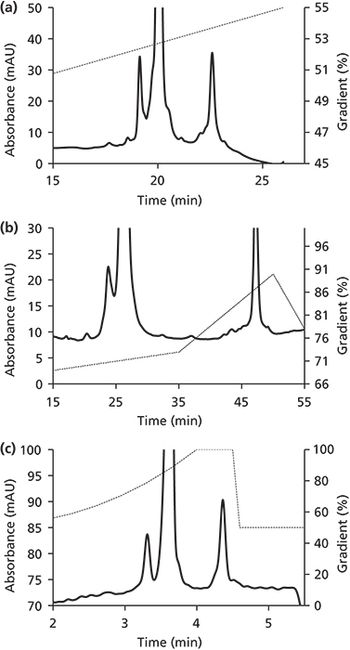
In this installment, we discuss fluorescence-detection size exclusion chromatography (FSEC) technology and its implications in protein characterization, specifically the development and characterization of biologics.

LCGC Europe
Fluorescence-detection size-exclusion chromatography and its implications in protein characterization, specifically the development and characterization of biologics, is reviewed.
LCGC Europe
In this article, we discuss the role of the gradient in protein separations by reversed-phase and ionexchange high performance liquid chromatography (HPLC).

LCGC North America
Isranalytica offered great talks on biotechnology, on topics from comprehensive protein characterization to middle-out proteomics to capillary electrophoresis for glycoprofiling.

LCGC North America
An overview of high-throughput process development (HTPD) tools and approaches, as well as a case study

LCGC North America
The authors discuss the specifics of analytical method validation.

LCGC North America
Glycoproteins appear to have become the most common biopharmaceutical product today, and they also seem to be increasing in popularity and importance with time.

LCGC North America
The authors provide an introduction to the topic of biosimilars, review the available regulatory guidance, and discuss the various scientific challenges that are present.

LCGC North America
In the first installment of LCGC's newest column, the authors address the topic of analytical biotechnology.

LCGC North America
The authors present the first installment in their examination of the basics of method validation.

LCGC North America
The authors explore how true (complete) method validation of biopharmaceuticals must be forthcoming.

LCGC North America
The guest columnists continue their examination of how statistically rigorous QbD principles can be put into practice.

LCGC North America
This second part of the series describes the data loss inherent in most early method development experiments due to coelution, peak exchange, and the general difficulty of accurately identifying peaks across the experiment trial chromatograms.

November 1st 1999

June 1st 2001

June 1st 2011

August 1st 2010

January 1st 2011