LCGC International Column Editor Michael Dong concludes his three-part series on HPLC fundamentals for practicing scientists with a case study concerning pharmaceutical development using UHPLC.

LCGC International Column Editor Michael Dong concludes his three-part series on HPLC fundamentals for practicing scientists with a case study concerning pharmaceutical development using UHPLC.

HPLC is indispensable in pharmaceutical analysis and quality control despite some notable shortcomings in laboratory testing and processes to ensure regulatory compliance.

This review highlights the high performance liquid chromatography (HPLC), mass spectrometry (MS), chromatography data systems (CDS), and related products that were introduced from 2024 to 2025, summarizing the technical specifications of each product and highlighting significant user benefits.

Michael Dong recently discussed sample preparation (SP) techniques for small-molecule drug substances and solid dosage forms.

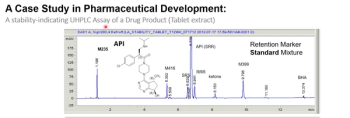
This paper describes the content of a well-written analytical procedure for regulated high-performance liquid chromatography (HPLC) testing. A stability-indicating HPLC assay for a drug product illustrates the required components for regulatory compliance, including additional parameters to expedite a laboratory analyst’s execution.

This article provides a high-level overview of Current Good Manufacturing Practice (cGMP) regulations and related public quality standards.

What you need to know about pharmaceutical regulations as an analytical chemist is provided here, including an explanation about how working in a Good Manufacturing Practice (GMP) environment can yield positive results.

An examination of the new high performance liquid chromatography (HPLC), mass spectrometry (MS), chromatography data systems (CDS), and related products that were introduced over the last year.

This case study of the career path of a separation scientist illustrates the diversified roles of an analytical chemist in nonclinical drug development.

An overview of modern approaches to small-molecule drug discovery.

We kick off a new article series that offers an insider’s view of the pharmaceutical industry, focusing on the role of separation science and separation scientists.

Our annual review of new high performance liquid chromatography instruments, mass spectrometry systems, and data processing software.

With a new free simulator, educators and students can perform virtual HPLC experiments under isocratic or gradient conditions applicable to real instrumentation and method development.

This concise yet comprehensive overview of sample preparation for bioanalysis looks at sample preparation fundamentals, best practices, and modern trends—all illustrated with a case study.

This is the first article in a four-part series exploring the quantitative assessment of drugs and their metabolites in biological fluids (such as blood, plasma, and urine) and tissue homogenates using liquid chromatography–mass spectrometry (LC–MS).

Highlights of the new high-performance liquid chromatography, mass spectrometry, and chromatography data systems introduced over the past year.

In the pharmaceutical industry, method validation is essential. But what are the best practices? We review regulatory requirements, validation parameters, methodologies, acceptance criteria, trends, and software tools.

Here we provide an overview of the fundamentals and best practices on the development of stability-indicating HPLC methods for drug substances and products. We explain both traditional and easier modern approaches to developing stability-indicating HPLC methods—including using a universal generic method for new chemical entities—and address regulatory considerations and life cycle management strategies.

Determining product shelf life is a regulatory requirement for pharmaceuticals and many other regulated consumer products. In this comprehensive overview of stability studies and testing, we summarize current regulatory requirements, share industry practices for forced degradation, and explain approaches for reduced testing and data evaluation to expedite stability study timelines.

Our annual review of new high performance liquid chromatography and mass spectrometry instruments, chromatography data systems (CDS), and related products, including a summary of their significant features and user benefits.

This technical overview of chromatography data systems (CDS) looks at how CDS are designed, how they can be used most effectively, and what developments we can expect in the future.

The authors provide a technical overview of the design and operating principles of variable wavelength and photodiode array detectors, and include historical perspectives and common practices in operation and maintenance.

We explain recent trends in HPLC autosampler design, provide recommendations for selecting one, and offer guidelines for operation and troubleshooting.

New HPLC and MS systems: A review of instrumentation trends and the current market along with new instruments, modules, chromatography data systems, and related software.

Modern trends in LC mobile-phase selection and preparation include using simpler mobile phases, increased use of MS-compatible mobile phases, and eliminating filtration and certain mobile-phase additives. Here, we discuss those and other trends and best practices, as well as the fundamentals behind them.

These ten propositions are widely acknowledged, but frequently neglected, by practitioners of high performance liquid chromatography (HPLC).

Comprehensive characterization of ADCs requires increasingly powerful approaches consisting of small- and large-molecule techniques.

An overview and assessment of the latest HPLC and mass spectrometry instruments, modules, chromatography data systems, and related software.

What are the potential problems that may be encountered using UHPLC systems and methods, and what strategies can be used for their mitigation?

UHPLC’s benefits include fast analysis, high-resolution separations, reduced solvent and sample usage, enhanced sensitivity and precision, and more.