LCGC Asia Pacific
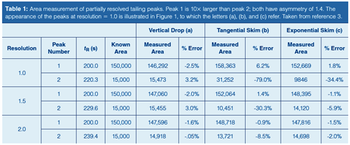
Separation scientists may seek an optimum spot between chromatographic performance required to obtain sufficient results quality, and the time and resources needed to do so. This instalment of “GC Connections” examines the factors that control peak resolution-one of the main drivers of separation quality-and how chromatographers can use this information to find an optimum between time, cost, and performance.



