
LCGC Asia Pacific
Application Note

LCGC Asia Pacific
Application Note

LCGC Asia Pacific
In contrast to isocratic separations, gradient elution can sometimes seem to be counter-intuitive. Yet with the proper perspective, the things we intuitively understand about isocratic methods apply in a similar manner to gradients.

LCGC Asia Pacific
Application Note
LCGC Asia Pacific
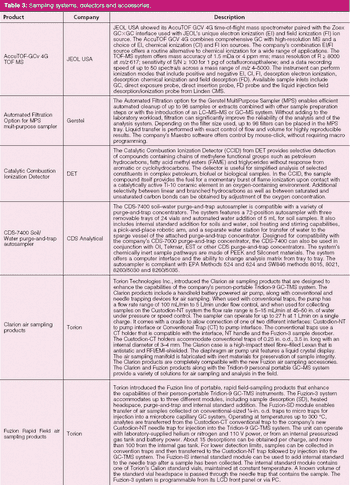
The first instalment of the Perspectives in Modern HPLC column highlights some of the new HPLC systems, accessories and related technology introduced at Pittcon this year.
LCGC Asia Pacific
John Hinshaw presents his annual review of new developments in the field of gas chromatography introduced at Pittcon 2013.

LCGC Asia Pacific
The removal of azide as a potential impurity from a drug substance may be critical to its safety profile. The quantitation of this impurity is an important control parameter. This article describes a simple and practical assay for azide using chemical derivatization and HPLC. The method is shown to be suitable for the intended purpose on three example test materials. Potential issues for wider applications are discussed.

LCGC Asia Pacific
Click the title above to open the LCGC Asia Pacific June 2013 regular issue, Vol 16, No 2, in an interactive PDF format.