- The Column-06-11-2018
- Volume 14
- Issue 6
Tips & Tricks: Aqueous GPC/SEC: Influence of Salt and pH
While gel permeation chromatography/size-exclusion chromatography (GPC/SEC) of uncharged molecules in organic eluents in most cases is a straightforward task, aqueous GPC/SEC of polyelectrolytes usually requires more parameters to be considered and optimized. This instalment of Tips & Tricks explains more.
While gel permeation chromatography/size-exclusion chromatography (GPC/SEC) of uncharged molecules in organic eluents in most cases is a straightforward task, aqueous GPC/SEC of polyelectrolytes usually requires more parameters to be considered and optimized. This instalment of Tips & Tricks explains more. -Wolfgang Radke, PSS Polymer Standards Service GmbH, Mainz, Germany
Gel permeation chromatography/sizeâexclusion chromatography (GPC/SEC) is the workhorse in macromolecular science when it comes to determination of molar masses and molar mass distributions. Once a suitable method is established, the simple methodology, the low workload needed for sample preparation, and the possibility to automate the analysis allows large numbers of samples to be analyzed unattended, thus making the process cost-effective.
However, while identifying appropriate combinations of stationary and mobile phases is often straightforward when using organic eluents, method development of aqueous GPC/SEC-particularly for macromolecules bearing ionizable groups (polyelectrolytes)-is usually more demanding.
Why do Polyelectrolytes Require the Addition of Salts?
Polyelectrolytes are classified as polycations or polyanions. In an aqueous environment, they dissociate to different degrees, depending on pK, pH, and salt concentration. If a polyelectrolyte is dissolved in pure water, only a certain fraction of the repeating units will be dissociated, resulting in charges along the polymer chains and free counter ions (1 in Figure 1). However, complete dissociation will rarely occur because it would require building up a huge charge around the macromolecule.
The charges along the chain will repel each other, resulting in stretching and coil expansion of the polymer chain (2 in Figure 1), which is associated with a considerable increase of the viscosity of the solution. This is known as the “polyelectrolyte effect”.
In GPC/SEC experiments the large size, resulting from increased coil expansion of a polyelectrolyte in a salt-free eluent, will prevent the polyelectrolyte chain from entering smaller pores. Consequently, the polyelectrolyte exhibits a much lower elution volume as a non-ionized sample of similar chain length. If the molar mass of a polyelectrolyte is derived from a calibration curve established using uncharged water-soluble polymers (for example, polyethylene glycols or pullulans) very different molar masses will result.
To counteract the expansion of the polyelectrolyte chain, low molar mass salts are usually added to shield the electrostatic interaction along and among the chains (3 in Figure 1). Upon addition of a salt, the electrostatic repulsion along the chain is shielded and the polyelectrolyte chain shrinks strongly, resulting in a severe change in elution volume.
An example is shown in Figure 2 where the chromatograms of low molar mass polyethylene imines (PEI) are compared using salt-free and salt-containing eluents. PEIs can be conveniently analyzed using acetic conditions. In aqueous trifluoroacetic acid without the addition of NaCl the sample elutes at a significantly lower elution volume compared to the salt-containing eluent. In contrast, the elution volumes of pullulan standards are not altered significantly by the addition of salt. Therefore, if the two chromatograms are evaluated using a pullulan-based calibration curve, large differences in molar mass will result. For the example shown, the molar mass derived in the salt-free eluent exceeds the molar mass in the salt-containing solution by approximately one order of magnitude.
It should be mentioned that even small salt concentrations significantly alter the size of polyelectrolyte chains. It is therefore hard to establish stable chromatographic conditions for polyelectrolytes in the absence of salt and so it is advisable to add salt to aqueous eluents to obtain stable results.
In most aqueous applications salt addition is necessary to shield electrostatic interaction along and among the macromolecules as well as between the macromolecule and the stationary phase. In addition to coil expansion from electrostatic interaction of charges along the polymer chains, interactions of charges with the stationary phase could also occur. Such ionic interaction will be shielded by the addition of salt as well. Therefore, pure water is rarely applied as eluent in aqueous GPC/SEC.
Why is the pH Important?
The pH of the eluent is another major factor that influences elution in aqueous eluents. The extent of dissociation and, thus, the polarity of the macromolecule, for example, of a poly(acrylic acid), depends on pH. At high pH a significant fraction of the carboxylic acid groups is neutralized resulting in a copolymer containing carboxylic acid and carboxylate groups, while at low pH, dissociation is strongly suppressed, resulting in a less polar poly(acrylic acid). Depending on the polarity of the stationary phase, nonpolar interaction between the less polar poly(acrylic acid) and the stationary phase could occur in aqueous eluents, resulting in late or even no elution at all from the column. This explains why polyacrylic acid in phosphate buffer at pH 9 elutes with high reproducibility as a wellâshaped peak, while no elution from the same column is observed in phosphate buffer at pH = 5. Consequently, a suitable pH is usually required to properly elute charged polymers.
Changing pH does not only change dissociation of the polyelectrolyte, but could also alter the charge state of the stationary phase itself if the stationary phase contains ionizable groups. The pH of the eluent applied, which in turn depends on the chemical structure of the polymeric analyte, influences the type of column material to be chosen.
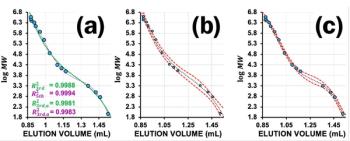
If at a given pH the macromolecule and the stationary phase contain similar charges, the macromolecule will be repelled from some part of the pore. This might ease counterbalancing undesired nonpolar interaction with the stationary phase. However, the repulsion of the macromolecule from the stationary phase results in an apparent reduction of the pore size of the stationary phase. Despite that, one might still observe typical GPC behaviour for the given polyelectrolyte structure, that is, larger polyelectrolytes eluting before the smaller ones. However, as neutral molecules of the same size have a larger accessible pore volume because of the absence of the above described repulsive forces, the separation is not strictly based on size as indicated in Figure 3(a). To reestablish a true size-based separation, the electrostatic repulsion between the macromolecule and the stationary phase needs to be shielded, for example, by the addition of a low molar mass salt (Figure 3[b]).
Additional Recommendations for Method Development
Finally, if the water-soluble macromolecule contains additional nonpolar structures, the addition of organic modifiers might be required to reduce or eliminate undesired nonpolar interaction of the macromolecule and the stationary phase.
Summary
- Successful GPC/SEC in aqueous eluents requires careful consideration of the interaction of stationary and mobile phase with the macromolecule.
- pH changes the charge state of the analyte molecule, thereby influencing polarity of the macromolecules. As a result, adsorptive nonpolar interaction with the stationary phase might become dominant resulting in late or incomplete sample elution.
- In salt-free conditions, ionizable groups along the macromolecular chain repel each other, resulting in undesired coil expansion, giving the impression of the existence of very high molar mass products.
- Repulsive interaction of charges along the macromolecule as well as between the macromolecules or between the macromolecule and the stationary phase can be shielded by the addition of low molar mass salts.
Wolfgang Radke studied polymer chemistry in Mainz, Germany, and Amherst (Massachusetts, USA) and is head of the PSS application development department. He is also responsible for instrument evaluation and for customized trainings.
E-mail:
Website:
Articles in this issue
almost 8 years ago
Vol 14 No 6 The Column June 2018 North American PDFalmost 8 years ago
Vol 14 No 6 The Column June 2018 Europe and Asia PDFabout 8 years ago
Resources for Careers in Metabolomics for Early Career Researchersabout 8 years ago
Josef Heiland Receives Eberhard Gerstel Awardabout 8 years ago
Agilent Announces Genohm Acquisitionabout 8 years ago
LabConnect Expands