- The Column-12-10-2019
- Volume 15
- Issue 12
Tips & Tricks GPC/SEC: “Green” GPC/SEC and Solvent Alternatives
The “greenest solution” is certainly using no solvent but gel permeation chromatography/size-exclusion chromatography (GPC/SEC) as a liquid chromatography (LC) technique requires the use of a mobile phase. The growing awareness of the need for more sustainable (greener) solutions has focused attention on environmentally- and health-friendly solvents and solutions.
The “greenest solution” is certainly using no solvent but gel permeation chromatography/size-exclusion chromatography (GPC/SEC) as a liquid chromatography (LC) technique requires the use of a mobile phase. The growing awareness of the need for more sustainable (greener) solutions has focused attention on environmentally- and health-friendly solvents and solutions.
Traditionally gel permeation chromatography/size-exclusion chromatography (GPC/SEC) is a “slow” method that uses long columns with large diameters. Water-soluble macromolecules can, by their very nature, be analyzed under relatively “green” conditions, but for other macromolecules specific, environmentally challenging organic solvents are often required.
In contrast to high performance liquid chromatography (HPLC), where separations can be optimized by varying the solvent gradient or the temperature, GPC/SEC separations rely solely on the accessible pore volume. A minimum amount of material or pores is required to achieve sufficient resolution and therefore, compared to HPLC, longer columns with a larger diameter have to be used. To increase the pore volume (and thus the resolution), often several columns are combined to create column combinations or column banks (1).
A drawback of this approach is that analysis time and eluent consumption increases with the number of columns. Despite these very challenging experimental conditions, strategies have been implemented to reduce the solvent consumption (2). This includes software solutions such as overlaid injection and, if applicable, a reduction of particle size and the use of small particles in columns with smaller dimensions in optimized hardware.
Besides approaches for solvent reduction the chemical industry is also investigating complete substitution of the solvent. In some areas solvents from renewable resources or less harmful alternatives have already been successfully applied. For example, ethyl lactate, an ester of lactic acid, is a “green” solvent derived from processing corn. As a result of its high solvating power, it has replaced solvents such as toluene, acetone, or xylene in the polyurethane industry as a cleaner alternative, resulting in safer workplaces. Other greener solvent alternatives for traditional organic mobile phases are summarized in Table 1.
However, the availability of a greener alternative is not the only parameter to be considered when researching more sustainable GPC/SEC solvents. Prerequisites for the suitability of a GPC/SEC mobile phase are also that there is:
- a matching stationary phase
- conditions that allow for interaction-free size separation
- soluble calibration standards that allow for the creation of a calibration curve
- sufficient detection options.
Medium Polar Organic Mobile Phase
A relatively straightforward approach is available for all materials that are analyzed in medium polar solvents, such as dimethylformamide (DMF). Medium polar stationary phase materials for interactionâfree GPC/SEC have been developed for the polarity range of these solvents and have proven their applicability in the last few years.
With only a very few exceptions, samples that are soluble in DMF are also soluble in dimethylacetamide (DMAc). As DMAc is less harmful compared to
DMF, it is preferred for method development.
Another potential alternative is dimethyl sulfoxide (DMSO), a solvent for which no H and P warnings (hazard and precautionary warnings) are reported. However, it is a penetration enhancer, so skin contact should be avoided. When working with DMSO in GPC/SEC, higher temperatures (60–80 °C) are required as a result of the high viscosity of this mobile phase. Suitable calibration standards are polymethyl methacrylate (PMMA) and (depending on the stationary phase chemistry) polystyrene (PS).
Nonpolar Organic Mobile Phases
Tetrahydrofuran (THF) is still one of the most commonly used nonpolar organic mobile phases in GPC/SEC. Potential replacements for THF are ethyl lactate and 2-methyl THF.
Ethyl lactate is a colourless liquid with a discreet scent. Both PMMA and PS standards are soluble in ethyl lactate. To investigate the potential of this solvent as a THF substitute, a typical styreneâdivinlybenzene phase was transferred from THF to ethyl lactate. The transfer was straightforward and fast, and the plate count test confirmed the successful solvent exchange. Mixtures of PMMA and PS standards were then analyzed using a GPC/SEC system equipped with a refractive index (RI) detector. Figure 1 shows an overlay of a PS and a PMMA mixture, each comprising four different molar mass standards.
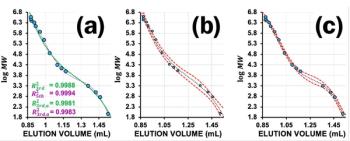
Although at first glance the chromatograms of PS and PMMA look promising, the elution of the PS standard of 1300 Da close to the RI system peak is a clue that a very thorough investigation is required. Lower molar masses of PS (oligomers) elute even with or after the typical RI system peaks. The overlay of the calibration curves (compare Figure 2) shows that the PMMA and the PS calibration curves differ strongly in the low molar mass region. Interaction of the PS oligomers in this mobile phase–stationary phase combination is the potential root cause for this observation.
Thus, only PMMA is a suitable calibration candidate when working with ethyl lactate on styrene-divinyl benzene materials, because PS most probably interacts with the stationary phase material.
2-Methyl THF was the second alternative to be tested. It is aIso a colourless liquid with a discreet scent. Again the two most common calibrants, PS and PMMA,
were soluble and could therefore be investigated further. The first chromatograms looked promising for both standard types, and additional tests were performed on a column combination with the highest resolution in the low molar mass area.
Figure 3 shows an overlay of the results for a mixture of four different molar mass standards of PS (green) and PMMA (orange). As a result of the very high resolution of the chosen column set in the low molar mass region, the lower (last eluting) PMMA and PS standards could be resolved into single oligomers resulting in multiple peaks.
PMMA and PS molar masses do not match 100%, which was expected because GPC/SEC separates based on hydrodynamic volume and not on molar mass. However, all peaks clearly elute before the total permeation limit and thus both substances are applicable for calibration.
As in the case of ethyl lactate, RI detection was used for the separations in 2-methyl THF. The difference in signal intensity for a comparable injected mass shows that PS has a higher dn/dc value and therefore larger response in 2-methyl THF compared to PMMA. This was less pronounced in ethyl lactate. However, in 2-methyl THF, detection with a UV detector is also possible for wavelengths above 230 nm.
Conclusions
- There are a few solvents that could be considered as “greener” organic solvents because they are produced from renewable resources or have less health risks.
- DMSO or DMAc are preferred over DMF for samples that can be analyzed in medium polar solvents.
- Alternatives for THF are currently being investigated. Stationary phases are suitable for the alternatives 2-methyl THF and ethyl lactate.
- Potential calibrants for 2-methyl THF are PS and PMMA. For ethyl lactate, only PMMA calibrations should be used when working on styrene-divinylbenzene stationary phases.
References
- D. Held, The Column9(22), 21–25 (2013).
- D. Held and J. Preis, The Column11(14), 12–16 (2015).
Daniela Held studied polymer chemistry in Mainz, Germany, and works in the PSS software and instrument department. She is also responsible for education and customer training.
Jasmin Preis studied polymer chemistry in Mainz and works in the PSS production department. She is responsible for custom synthesis of specialty polymers and particles.
E-mail:
Articles in this issue
over 6 years ago
Vol 15 No 12 The Column December 2019 Europe & Asia PDFover 6 years ago
Vol 15 No 12 The Column December 2019 North American PDFover 6 years ago
Peter de Boves Harrington Receives EAS Awardover 6 years ago
Instrumental Innovations 2019over 6 years ago
Investigating Primate Fertility Cues Using GC–MSover 6 years ago
Joel M. Harris Receives EAS Awardover 6 years ago
The 2020 LCGC Awards Winners are...over 6 years ago
Thermo Fisher Announce Owlstone Medical Collaborationover 6 years ago
How Does Your Laboratory Measure Up On Glassdoor?