Kanamycin and amikacin are aminoglycoside antibiotics used to treat serious bacterial infections. Amikacin is used for infections resistant to other aminoglycosides because it is less susceptible to enzymatic reactions.

Kanamycin and amikacin are aminoglycoside antibiotics used to treat serious bacterial infections. Amikacin is used for infections resistant to other aminoglycosides because it is less susceptible to enzymatic reactions.

Dionex - Iodide in Seawater and Saline Matrices

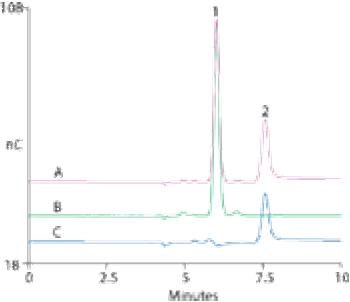
Oxytocin acetate samples were analysed successfully for both the peptide and acetate with a single method using a mixed-mode weak-anion-exchange column.
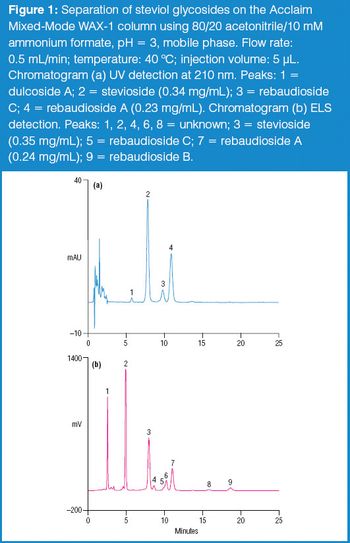
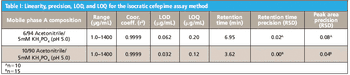
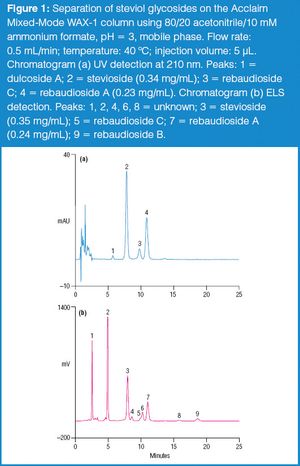
The stevia plant and its extracts have long been used as sweeteners in Asia and Latin America. Two steviol glycosides present in plant tissue, stevioside and rebaudioside A, are largely responsible for the sweet flavour.1 In December 2008, the US FDA placed rebaudioside A (also known as rebiana) on the Generally Recognized as Safe (GRAS) list of sugar substitutes to be used in foods, thereby allowing the use of rebiana as a commercial sweetener.2 The determination of steviol glycosides in these sweeteners is challenging due to their weak UV absorbance. Other detection methods, such as evaporative light scattering (ELS), can improve steviol glycoside quantification. In this proposed method, steviol glycosides were determined by UV and ELS detections in consumer sweeteners following separation on the Acclaim Mixed-Mode WAX-1 column.3
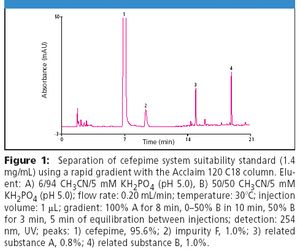
Development of cephalosporin antibiotics has led to compounds with a broad spectrum of activity against both Gram-positive and Gram-negative bacteria with low toxicity profiles. Cefepime, a fourth-generation cephalosporin, is a commonly prescribed broad spectrum antibiotic with improved activity against Gram-negative bacteria compared to other commercially available cephalosporins (1). Despite extensive research on this class of drugs, quantitative analysis and purity assays remain problematic (2).
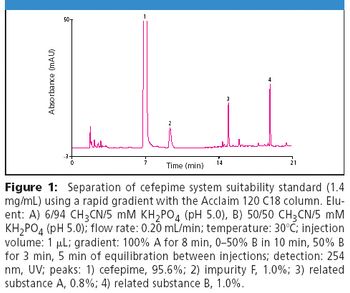
Cephalosporins contain a four-member β-lactam ring that is inherently strained and prone to hydrolysis and photolysis, limiting stability and leading to degradation products that may be toxic (1). In addition, synthetic byproducts are generated and persist during production of these antibiotics including cefepime. Analysis of cefepime purity is particularly challenging due to the presence of such isomeric synthetic impurities. The Acclaim® 120 C18, 3 μm can be used to meet and exceed the criteria set by the USP for determining related substances and assaying the purity of cefepime.

Published: June 1st 2011 | Updated:
Published: March 2nd 2010 | Updated:

Published: November 9th 2010 | Updated:

Published: April 1st 2010 | Updated:

Published: February 1st 2009 | Updated:
Published: September 1st 2008 | Updated: