Articles by Jeff S. Rohrer

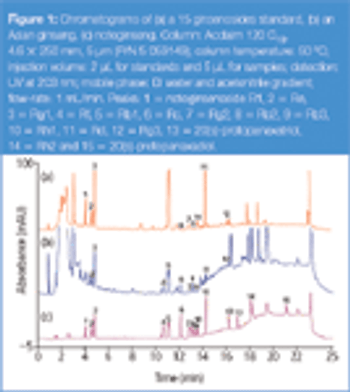
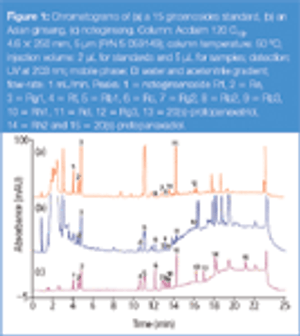
Glycoprotein monosaccharide compositional analysis is important for the development and manufacture of glycoprotein human therapeutics. Here we report a robust method for this analysis. This chromatographic method requires no mobile phase preparation thus saving time and delivering excellent retention time reproducibility that results in good method reproducibility.
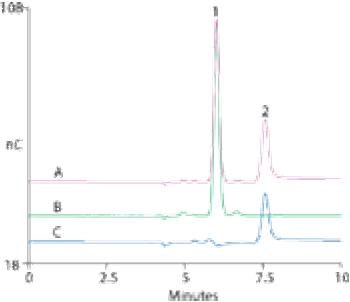
Kanamycin and amikacin are aminoglycoside antibiotics used to treat serious bacterial infections. Amikacin is used for infections resistant to other aminoglycosides because it is less susceptible to enzymatic reactions.
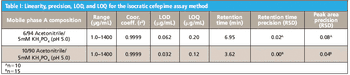
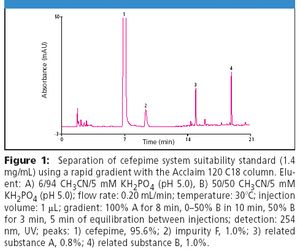
Development of cephalosporin antibiotics has led to compounds with a broad spectrum of activity against both Gram-positive and Gram-negative bacteria with low toxicity profiles. Cefepime, a fourth-generation cephalosporin, is a commonly prescribed broad spectrum antibiotic with improved activity against Gram-negative bacteria compared to other commercially available cephalosporins (1). Despite extensive research on this class of drugs, quantitative analysis and purity assays remain problematic (2).
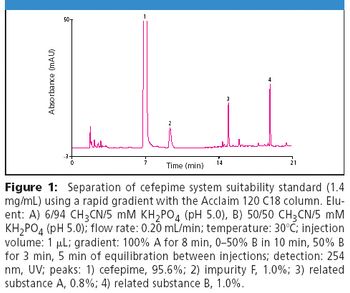
Cephalosporins contain a four-member β-lactam ring that is inherently strained and prone to hydrolysis and photolysis, limiting stability and leading to degradation products that may be toxic (1). In addition, synthetic byproducts are generated and persist during production of these antibiotics including cefepime. Analysis of cefepime purity is particularly challenging due to the presence of such isomeric synthetic impurities. The Acclaim® 120 C18, 3 μm can be used to meet and exceed the criteria set by the USP for determining related substances and assaying the purity of cefepime.

Urea and allantoin are added to cosmetic products for skin protection and regeneration, especially for the treatment of dry skin, and analyzed for QC purposes. As polar compounds, they are not ideal for reversed-phase HPLC separations. Neutral hydrophilic compounds like urea and allantoin are best analyzed by hydrophilic interaction chromatography (HILIC). Traditional HILIC columns use silica modified with a hydrophilic group such as diol or cyano. Analytes are adsorbed and subsequently eluted with mobile phases containing high percentages of organic solvent (>75%).