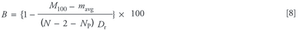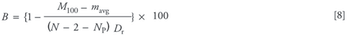
Protein unfolding and aggregation can be serious considerations when designing laboratory and preparative chromatographic purification steps. This problem has been studied most thoroughly within the contexts of reversed-phase chromatography and hydrophobic interaction chromatography. However, there are currently no robust methods for resin selection capable of predicting adsorbed-phase protein stability as a function of amino acid sequence, secondary or tertiary structure, or resin characteristics.