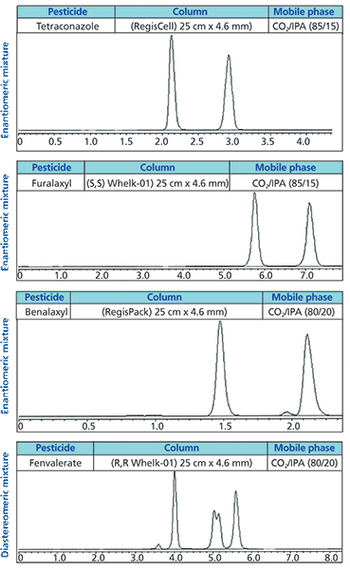
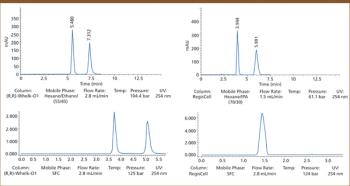
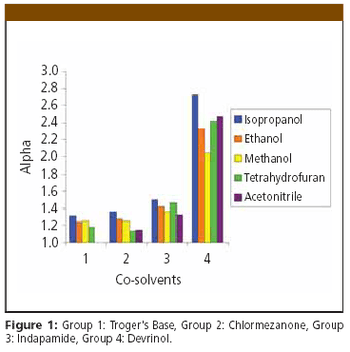
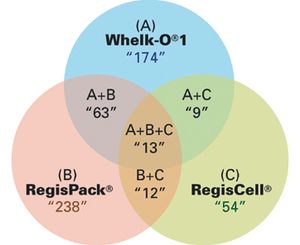
Successful therapeutic intervention often requires chiral medicines because of the intrinsic chirality of protein drug targets, which consist of L-amino acids. Potency, efficacy, and safety can be highly dependent on the precise stereochemical geometry of the molecules. Determining the biological profile of individual enantiomers in the early stages of drug discovery is important for successful optimization towards clinical candidates. Here we demonstrate the benefits of supercritical fluid chromatography (SFC) with three chiral stationary phases exemplified by high frequency resolution of 41 out of 50 chiral derivatives of eight commonly used drug discovery scaffolds including 1,3-thiazoles, 1,3-benzothiazoles, pyranoquinolones, indoles, and leucolines.