- LCGC North America-02-01-2017
- Volume 35
- Issue 2
Filters and Filtration in Liquid Chromatography—What To Do

Delivering samples to the analytical column in ‘clean’ mobile phase is important for robust methods and high quality results.
Editor’s Note from John Dolan: With this installment of “LC Troubleshooting,” you’ll begin to see evidence of a transition. I’m retiring as editor of this column in October 2017 and handing the reins over to Dwight Stoll. This is Dwight’s first article during the transition period-you’ll see more of him during the next few months. Many of you will recall that Dwight was the recipient of LCGC’s 2011 Emerging Leader in Chromatography award. He earned his Ph.D. from Peter Carr’s group at the University of Minnesota and now is tenured at Gustavus Adolphus College. I’ve had the privilege of knowing Dwight since his graduate school years and of working with him on a liquid chromatography (LC) column selectivity project. He’s one of the world’s leaders in two-dimensional LC (2D-LC), so I’m sure we’ll all gain some insights into the successful implementation of this exciting technique. Join me in welcoming Dwight onto theLCGC team.
Delivering samples to the analytical column in “clean” mobile phase is important for robust methods and high quality results.
Sometimes the instrumentation we use in LC works remarkably well in spite of our sometimes justified neglect of best practices designed to protect the instrument’s more-delicate components. One of the ways I balanced the checkbook during my graduate school days was doing instrument repair on nights and weekends. I vividly recall receiving an LC pump from a laboratory in Kansas that looked like it had sat out in the middle of a farm field through a Minnesota winter-the electronic boards were full of dust and the front area of the pump where the pump head sits was literally packed with dirt. To my amazement it still pumped liquid, albeit not with the accuracy we would expect.
For this installment of “LC Troubleshooting” I decided to focus on filters and filtering as they relate to proper, reliable operation of high performance liquid chromatography (HPLC) instruments. John Dolan has written extensively and often on these topics in the past (1–4), and yet many of the problems I discuss with LC practitioners in the field-and with students in my own laboratory-are in some way related to filters and filtering. Technologies, practices, and the demands of our laboratory settings change, too, and so this month I’d like to discuss what has stayed the same and what has changed, while providing some guidance for our everyday work.
In preparation for writing this column, I informally surveyed several people, because I was interested to know what folks are actually doing in their laboratories. I consider all of these people experts in LC, with an average of about 15 years of experience, and they represent a broad cross-section of our community, ranging from pharmaceutical and chemical companies to instrument manufacturers. What was strikingly evident from this survey was that people handle the topic of filters and filtering in diverse ways, even though instrument manuals and books on LC may suggest only one way. Our reality today is that the practice of LC is strongly influenced by a number of drivers that the manuals don’t consider, including minimizing the number of laboratory operations to minimize variation in analytical results, and optimizing the costs of analytical results from a holistic point of view (in other words, analyzing the benefit of component X to the method relative to the cost of the component).
The topic of filters and filtering is very important in LC because a number of components in a LC system are susceptible to premature failure if particulates are present in the mobile-phase flow path. For example, dust or other particulates in the mobile phase pulled from a solvent bottle can cause pump check valves to fail partially (they become leaky) or entirely (the pump produces no flow at all). Perhaps the most obvious failure mode involves particulates from injected samples or instrument components accumulating at the column inlet, resulting in a plugged column. In the following paragraphs I will discuss some of the most important points in an instrument setup and method related to filters and filtration. In each case I will describe best practices from the point of view of the instrument and will also discuss cases in which this advice may not be strictly followed for good reason.
We will see that many of these considerations boil down to thinking about risk and benefit in terms of the potential benefit derived from deviating from best practices and the risks associated with doing so.
Filtering to Protect the Pump
From the point of view of an LC pump, ideal solvents are absolutely free of particulate matter. Particulates can interfere with the sealing interface between the check valve ball and seat, causing it to leak or to fail entirely, resulting in inconsistent or no flow from the pump. Particulates can also accelerate wear on pump pistons and seals, which can result in leaks and flow-rate inaccuracy. These problems usually manifest themselves as noisy or irregular baselines, unexpected fluctuations in the operating pressure reported by the pump, and variations in retention time. Most sources of advice on this topic recommend that solvents should be filtered to 0.45 µm before use. Modern LC pumps come equipped with filters that are fitted to the inlet end of solvent lines that pull solvents from their reservoirs, but these filters have much larger porosity and are not designed to retain particles at the 0.5-µm level. Off-line filtration, however, adds cost (both consumables and operator time) and is a potential source of both chemical and particulate (yes, it happens) contamination of the solvent. Reputable manufacturers of solvents sold specifically for use in LC filter their solvents at the point of packaging (typically to 0.2 µm, and some to 0.1 µm), so one way to avoid in-house filtering altogether is to purchase and use premixed solvents. There was a time when choices for these solvents were limited to water and neat organic solvents, but these days most commonly used mobile phases can be purchased from major manufacturers as premixed solutions. For example, 0.1% formic acid in water, 0.1% formic acid in acetonitrile, and 0.1% ammonium acetate in water are all readily available. There is also an incredible array of premade buffers available for HPLC, including phosphate buffers, buffers with ion-pairing reagents added, and so on. The convenience associated with using these premixed solvents comes at a significant increase in purchase price over pure solvents, however, so users must decide if the increased cost is worth the benefits they offer. Some laboratories find that the increased cost of premixed solvents is offset by savings in labor and reduced downtime that would otherwise result from errors in mobile-phase preparation.
Mobile-Phase Additives
The majority of LC methods in use today involve some type of mobile-phase additive, including buffering agents and ion-pairing reagents. If we choose not to use the type of premade solvents described above, then we have to make a choice-do we filter the solvent after adding the additives, or not? At this point there is an opportunity to divide the question over two scenarios-those that involve liquid additives (for example, formic acid, ammonium hydroxide, and phosphoric acid), and those that involve solid additives (for example, potassium phosphate, sodium perchlorate, and sodium chloride). Among the experts I surveyed, very few reported that their laboratories filter solvents involving only liquid additives. I would report the same for my laboratory. The responses from the experts about solvents involving solid additives are mixed. A majority, but not all, of their laboratories routinely filter solvents containing solid additives. In my laboratory we often base our decision on the amount of solid additive used. If the amount of additive is small and it is highly soluble in the solvent-say 10 mM ammonium acetate in water-we don’t filter, but we always filter solutions containing high concentrations of additives. The compelling reasons to filter in these cases include increased risk of particulate contaminants present in the solid additive, particulates present due to incomplete dissolution of the additive, and, in the case of buffer–organic solvent mixtures, precipitation of the additive upon addition of the organic solvent.
Why or Why Not?
In the face of these compelling reasons to filter, especially when solid additives are used, one might reasonably ask-why not? Here is a short list of reasons, the significance of which often depends on context.
- Cost of filtering versus costs resulting from pump problems: If the risk of a solvent-related problem associated with the pump can be minimized through use of liquid additives and high-quality reagents, the cost associated with in-house filtering may outweigh costs associated with pump down-time. On the other hand, if your laboratory is a 24/7 operation and pump robustness is critical, then you may choose to prioritize in-house solvent filtering to maximize instrument up-time.
- Risk of introducing chemical and biological contaminants during filtering: These days very high quality membrane filters are available for filtering LC solvents that are unlikely to contaminate the solvent themselves. But, the filter is only one component of the filtration process. The rest of the filtration apparatus must be cleaned properly to avoid carryover of particulate and chemical contaminants to the filtered solvent. This approach is most certainly possible, but it is dependent on well trained operators, standard operating procedures, and a laboratory culture of respect for cleanliness. This issue of chemical and biological contaminants is especially important for methods focused on trace-level determinations, because the contaminants will be observed as noise or interferents when using very sensitive detectors (5,6).
- Fewer steps equal less opportunity for mistakes: One of the themes that emerged from my survey of experts is that there is a continuous drive for simplification of analytical procedures. If a step can be eliminated without compromising the quality of the analytical result, it is because fewer steps tend to translate into lower variability of results.
Microbial Contamination
Finally, the topic of microbial growth in solvent reservoirs is important. If microbes begin growing in the solvent container feeding the pump, there is potential for the instrument to be contaminated both physically and chemically. Here, too, some themes emerged from the experts I surveyed. The following best practices will help minimize problems with microbial growth, which is limited to aqueous solutions:
- Resist the temptation to top off solvent containers by adding new buffer to old buffer. Topping off provides a path for cross-contamination of buffer batches-even if the new buffer is perfectly sterile, it will be contaminated immediately if added to a solvent reservoir containing old buffer. Avoiding this practice adds some waste when the last drops of old buffer are thrown away, but this minor waste is a small price to pay for avoiding cross-contamination.
- For buffered aqueous solutions, particularly those near neutral pH, prepare concentrated stock buffer solutions (such as a solution containing a 10× concentration of buffering agent), refrigerate them upon storage, and dilute small quantities for use when needed. Prepare only enough of the working buffer to last one or two days to limit the time available for microbes to grow at room temperature.
- Add some antimicrobial agent to the buffer. Historically sodium azide was used for this purpose at low concentration (~ 0.05%), but this practice is becoming less common and many laboratories are choosing to simply add a small amount (many labs use 10%, but the “right amount” is case specific) of organic solvent such as acetonitrile to the buffer for this purpose.
Filtering to Protect the Column
The size of the interstitial spaces between particles of packed columns typically used in LC is a small fraction of the diameter of the particles themselves. Thinking of it this way, it becomes evident that the packed bed itself can act as an effective filter; clearly we want to employ the column for separation, not as a filter. The frits on the ends of the columns serve two purposes then: They retain the particles inside the column, and they prevent particulates carried by the mobile phase from entering and partially blocking flow through the particle bed. Accumulation of particulates on the frit at the inlet side of the column can also lead to plugging of the frit, rendering the column unusable because of the increased pressure drop over the frit. In the preceding section I discussed the mobile phase as it is drawn into the pump as a source of particulates. But even if the mobile phase entering the pump is perfectly particulate-free, there are several potential sources of particulates entering the flow stream between the pump and the column. Most prominent among these are shedding of pump-seal material (chunks of polymer); shedding of valve rotor seals (again, chunks of polymer), including the sample injection valve and column switching valves; and the sample itself. The topic of particulates in the sample is one that clearly crosses over into the area of sample preparation, and one could easily write an entire column on this topic alone. Here we will focus on the first two potential sources, the instrument-related sources of particulates.
In-Line Filters VersusGuard Columns
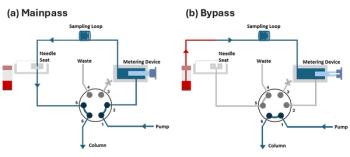
In the context of a LC instrument, an in-line filter is usually one that is placed in the mobile-phase flow path at some point between the pump outlet and the column inlet. These filters have evolved over the years to the point that the most high performing ones can be used at very high pressures, have small throughpore sizes (down to 0.2 µm), and have internal volumes in the microliter range so as to limit contributions to peak broadening. The sole purpose of these filters is to collect particulates in the mobile phase as a means of preventing damage to downstream components from these particulates. In my laboratory we do not regularly use in-line filters immediately after the pump, but we are very disciplined about using them between valves with polymeric rotor seals (for example, autosampler and column-switching valves) and the analytical column. We find that these seals shed a lot of particles as they wear, and that capturing them with an in-line filter generally increases the lifetimes of our analytical columns and small diameter capillary connection tubing. I would say that only about 50% of the LC instruments I see in a year in different laboratories have an in-line filter installed ahead of the analytical column, which is very surprising to me. Replacement filters cost anywhere from a few dollars to $25 depending on their features, but it is easy to justify replacing a few of these if we can significantly increase the lifetimes of our analytical columns and improve instrument robustness in general. It is important to note that some applications involving very small peak volumes (for example, small particle sizes and short columns) will be more susceptible to peak broadening inside the in-line filter. In these cases one has to think carefully about which is more important-minimizing dispersion, or protecting the column from particulates.
Guard columns are essentially very short versions of the analytical column (often with the same packing material and diameter, but lengths on the order of 5 or 10 mm) that are screwed into the column inlet. One can argue that guard columns can also serve as particulate filters to protect the analytical column, but this approach is a much more expensive way of doing so (on the order of 10–20% of the column purchase price per guard column). Instead, guard columns are more appropriately used for chemical protection of the analytical column. For example, sample constituents that are very highly retained by the analytical column, or perhaps will not even be eluted under the conditions of the experiment, can be trapped by the guard column. The contaminated guard column can then be replaced with a new one after the analysis of some number of samples at a fraction of the price of a new analytical column. The experts I surveyed reported more frequent use of in-line filters than guard columns, citing the cost of guard columns as being generally too high to justify their use in a uniform way.
Closing Thoughts
In this installment of “LC Troubleshooting,” I made generalizations where possible about when and why to filter. Of course there are special situations where these generalizations are not applicable. For example, certain types of detectors, such as light-scattering detectors used for polymer analysis, are very sensitive to particulates in the mobile phase and may require more-thorough and more-consistent filtering of mobile-phase solvents. LC users should think carefully about special requirements associated with their particular analyses and adjust their approaches to the use of filters and filtering accordingly.
References
(1) J.W. Dolan, LCGC North Am.28, 28–30 (2010).
(2) J.W. Dolan, LCGC North Am. 26, 352–355 (2008).
(3) J.W. Dolan, LCGC North Am.3, 956–960 (1985).
(4) J.W. Dolan and V.N. Berry, LCGC N. Am.1, 542–544 (1983).
(5) B.O. Keller, J. Sui, A.B. Young, and R.M. Whittal, Anal. Chim. Acta. 627, 71–81 (2008). doi:10.1016/j.aca.2008.04.043.
(6) S. Williams, J. Chromatogr. A.1052, 1–11 (2004). doi:10.1016/j.chroma.2004.07.110.
Dwight Stoll and John Dolan are the coeditors of LC Troubleshooting, and Stoll is the author of this installment.
Dwight Stollis Associate Professor and Co-Chair of Chemistry at Gustavus Adolphus College in St. Peter, Minnesota. He has authored or coauthored 48 peer-reviewed publications in separation science and more than 90 conference presentations. His primary research focus is on the development of two-dimensional liquid chromatography (2D-LC) for both targeted and untargeted analyses. He has made contributions on the topics of stationary-phase characterization, new 2D-LC methodologies and instrumentation, and fundamental aspects including reequilibration in gradient elution reversed-phase LC and analyte focusing. He is the 2009 recipient of the John B. Phillips Award for contributions to multidimensional gas chromatography, the 2011 recipient of LCGC’s Emerging Leader in Chromatography Award, and the 2015 recipient of the American Chemical Society Division of Analytical Chemistry Award for Young Investigators in Separation Science. Direct correspondence about this column via e-mail to
Articles in this issue
about 9 years ago
Advantages of Using Nitrogen in Capillary GCabout 9 years ago
Identifying and Discovering Marine Lipid Biomarkers Using LOBSTAHSabout 9 years ago
LCGC’s New “Go-To” Guyabout 9 years ago
Going Through Phasesabout 9 years ago
The 2017 LCGC Awardsabout 9 years ago
Vol 35 No 2 LCGC North America February 2017 Regular Issue PDFNewsletter
Join the global community of analytical scientists who trust LCGC for insights on the latest techniques, trends, and expert solutions in chromatography.