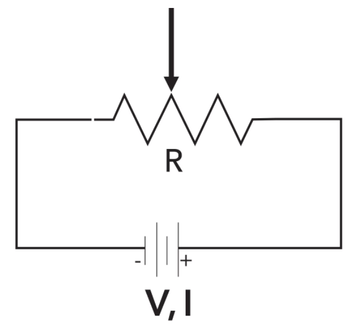
Computers control all aspects of modern GC instrument operation, from temperature to valve actuation. We look under the hood to see how this works.

Computers control all aspects of modern GC instrument operation, from temperature to valve actuation. We look under the hood to see how this works.
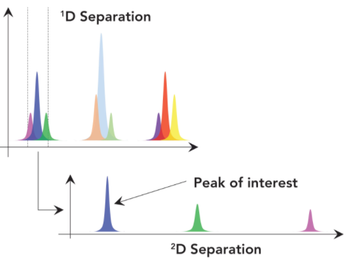
In 2D-LC, properties of the mobile phase used in first step can negatively affect the second step. We explain why this problem happens and how to avoid it.

IMS is a valuable tool for biopharmaceutical analysis. Two formats in particular have proven useful: cyclic IMS and structures for lossless manipulations (SLIM).

In the pharmaceutical industry, method validation is essential. But what are the best practices? We review regulatory requirements, validation parameters, methodologies, acceptance criteria, trends, and software tools.

Data systems are important in ensuring data integrity, but what should laboratories do beyond looking at data systems—with respect to people, processes, and culture? To understand more, we spoke to Mark Newton, of Heartland QA, in Lebanon, Indiana, in a podcast interview.

Consumer marketing approaches are creeping into the marketing of scientific instruments. With a careful approach, you can cut through the hype.
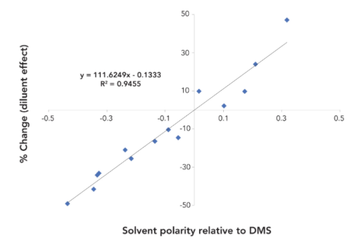
In static headspace gas chromatography (HS-GC) sample diluents and sample matrices can affect analytical method sensitivity, accuracy, and interferences. By applying the rules revealed by this study, these problems can be avoided.