LCGC North America
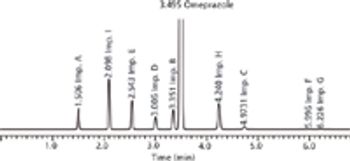
Quality-by-design principles are applied to build in a more scientific and risk-based multifactorial strategy in the development of a UHPLC method for analyzing a drug and its related impurities.

LCGC North America
Quality-by-design principles are applied to build in a more scientific and risk-based multifactorial strategy in the development of a UHPLC method for analyzing a drug and its related impurities.

LCGC North America
The selective removal of a fat substitute in food products is discussed to demonstrate options for obtaining selectivity during extraction.

LCGC North America
Key considerations for setting up or troubleshooting a GC method.

LCGC North America
A discussion of the future role of miniature MS systems, the need for simplification in operation, the role of ambient ionization, and challenges in development and commercialization.

LCGC North America
The analysis of pesticide residues in food samples from the state of Connecticut's regulatory monitoring program are compared to USDA and US FDA results.

LCGC North America
What kind of adjustments need to be made when scaling an isocratic method?


LCGC North America
Click the title above to open the LCGC North America February 2014 regular issue, Vol 32 No 2, in an interactive PDF format.