- May 2021
- Volume 17
- Issue 05
Driving Under the Influence of Drugs: PESI for the Measurement of Illicit Drugs in Saliva
The benefits of a PESI-MS/MS approach to detect illicit drugs in saliva.
This article demonstrates how a new probe electrospray ionization tandem mass spectrometry (PESI-MS/MS) method can be used to measure illicit drugs in saliva, confirming positive tests for driving under the influence of drugs (DUID) in France. A new method was developed for the measurement of illicit drugs in saliva using a PESI-MS/MS approach. Satisfactory results were obtained for eight opiates, amphetamines, and cocaine derivatives.
Probe electrospray ionization (PESI) is an ambient ionization method where a disposable solid needle is used as a sample probe, and electrospray ionization (ESI) is used as an emitter. Coupled with a mass spectrometer, it allows very fast analyses with minimal sample preparation. Unlike PESI, traditional mass spectrometry (MS) ion sources have to be maintained in a vacuum, and sample preparation is more extensive.
In France, confirmation of a positive test for driving under the influence of drugs (DUID) is performed in an authorized laboratory using a liquid chromatography (LC)–MS/MS method. How can a new method be developed for the measurement of illicit drugs in saliva using a PESI-MS/MS approach?
The following conditions were defined: 50 μL of saliva was mixed in a methanol–ethanol–ammonium formate buffer. After adding deuterated internal standards, a 10 μL measure was set in the PESI system. A scheduled multiple reaction monitoring (MRM) approach with two transitions per compound was developed. The total run time was about 1.5 min. Based on ISO
15189, validation steps included the determination of the limit of detection
(LOD) and the limit of quantification (LOQ), as well as repeatability and reproducibility analysis.
Satisfactory results were obtained for eight opiates, amphetamines, and cocaine derivatives. LODs in particular were below the limits defined by the law (10 ng/mL). In a sample of 52 real cases, the new method yielded results comparable to those of the LC–MS/MS method routinely used in the laboratory.
It is feasible to use PESI-MS/MS for a very fast measurement of illicit drugs in saliva. Further analytical improvements are needed to integrate all the compounds required by law.
Illicit Drug Analysis for Drivers
Usually, when a driver is suspected of consumption of illicit drugs, roadside testing is performed with an immunological kit based on an oral fluid (OF) or a urine sample. If positive, this test has to be confirmed in a second sample by a gold standard method, such as LC–MS/MS.
The legislation of most countries concerning DUID is based on impairment (“effect of the drug”) or on analytical limits (“cut-off” approach) (1). In some countries, including France, this confirmation is now typically (almost systematically) performed using an OF sample, and the regulatory approach does not apply a per se law (such as the driver is sentenced if the drug level is above a predefined limit), but based on a zero tolerance law (that is, driving with a measurable amount constitutes an offense). Consequently, laboratories have to detect a list of illicit drugs at a minimum concentration.
In France, for example, this “cut-off” is 10 ng/mL for cocaine, benzoylecgonine (BZE), morphine, 6-monoacetylmorphine (6-AM), amphetamine, methamphetamine, 3,4-methylenedioxy-N-ethylamphetamine (MDEA), 3,4-methylenedioxyamphetamine (MDA), and 3,4-methylenedioxy-Nmethylamphetamine (MDMA) (2).
What is PESI?
PESI is an ambient ionization method. It was developed by Kenzo Hiraoka of the University of Yamanashi, Japan, who published the first article on the subject in 2007 (3). Briefly, it contains a disposable solid needle used as a sample probe and an electrospray ionization (ESI) that is used as an emitter (3). This probe needle repeatedly moves down (that is, “dives” into the sample) and then moves up when ionization occurs. The compounds are ionized periodically and their resultant ions are pushed into the MS/MS system.
Consequently, when coupled with a MS system, the PESI makes a direct analysis of liquid samples and solid samples possible without pre‑treatment.
Publications and Applications of the PESI Technique
Different applications of the PESI technique have been reported in clinical toxicology, forensic science, and metabolic profiling. Zaitsu et al. applied PESI to measure 26 endogenous metabolites of mice liver, including amino acids, organic acids, and sugars (4). The same team also applied PESI to monitor eight cerebrum metabolites related to central energy metabolism in an isoflurane‑anesthetized mouse (5).
Johno et al. proposed an automatic diagnosis system of atherosclerotic disorders by measuring metabolites including cholesterol sulphate and a phospholipid, both promising new biomarkers of atherosclerosis (6). Usui et al. applied the PESI technique with LC and triple quadrupole (QQQ)-MS/MS for the detection of acetaminophen in less than 2 min (7). The same team also proposed a similar approach for the diagnosis of acute intoxications to glyphosate (8).
A PESI-MS/MS Method for Illicit Drugs
In this study, a PESI-MS/MS application for the determination of some illicit drugs in OF was developed. From a practical point of view, the main objective was to obtain performances in accordance with French legislation as described previously. The principle of the method can be summarized as follows:
50 μL OF samples were diluted in 450 μL of a 25:25:50 methanol–ethanol–ammonium formate 10 mM buffer. In a previous paper, Usui et al. (7,8) proposed an ethanol–ammonium formate buffer, but in preliminary studies it was observed that either methanol or ethanol could be necessary depending on the molecule.
From this 500 μL, 180 μL was mixed with 20 μL of a solution containing eight deuterated internal standards at 50 ng/mL. Finally, a 10 μL sample of diluted OF was placed on the dedicated plastic sample plate and set in the PESI ion-source.
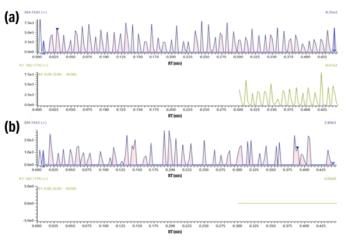
LC–QQQ-MS/MS with a PESI ion source was used for the determination of cocaine, BZE, morphine, 6-MAM, methamphetamine, MDEA, MDA, and MDMA. A scheduled MRM approach with simultaneous detection of two transitions of two compounds, acquired in the positive mode, was developed (Figure 1). For an MRM approach, three transitions for each compound are usually recommended. However, by increasing the analysis time, desolvatation of the solvents was observed.
The total run time was 1.31 min for detection of the eight compounds. The whole procedure was validated according to the EMEA guidelines (9). A summary of performances is given in Table 1.
Along the validation process, hundreds of OF samples free of illicit drugs were analyzed. Particular matrix effects for the eight molecules could not be observed. A typical extracted ion count of morphine, obtained for an OF sample spiked at 10 ng/mL, is presented in Figure 2.
The overall procedure was applied to 52 real cases of DUID that were also analyzed by a validated LC–MS/MS method. Briefly, the method is routinely used by the laboratory of Limoges University, France (accredited according to ISO 15189) and uses a QuEChERS sample preparation and LC–QQQ–MS/MS (10).
Seventy-three molecules were detected in these 52 samples: MDMA (n = 17), MDA (n = 1), cocaine (n = 26), BZE (n = 22), 6-MAM (n = 4), and morphine (n = 3). Each compound was systematically detected with both methods and excellent correlations were calculated between the new PESI-MS/MS method and the classical LC–MS/MS method. This study is illustrated in Figure 3, which reports the regression analysis for cocaine, BZE, and MDMA.
Conclusion
These experiments demonstrate the feasibility of using a PESI-MS/MS approach for the measurement of illicit drugs in saliva. With this approach, the detection is performed in about 1.5 min without an extraction procedure. The current procedure includes only eight molecules, and further studies are needed to increase the number of illicit drugs that could be tested in a single run.
References
- A.W. Jones, J.G. Morland, and R.H. Liu, Forensic Sci Rev. 31(2), 103–140 (2019).
- https://www.legifrance.gouv.fr/eli/arrete/2016/12/13/AFSP1636875A/jo. Accessed 10 March 2021.
- K. Hiraoka, K. Nishidate, K. Mori, D. Asakawa, and S. Suzuki, Rapid Commun. Mass Spectrom. 21(18), 3139–3144 (2007).
- K. Zaitsu, Y. Hayashi, T. Murata, T. Ohara, K. Nakagiri et al., Anal. Chem. 88(7), 3556–3561 (2016).
- K. Zaitsu, Y. Hayashi, T. Murata, K. Yokota, T. Ohara et al., Anal. Chem. 90(7), 4695–4701 (2018).
- H. Johno, K. Yoshimura, Y. Mori, T. Kimura, M. Niimi et al., Metabolomics 14(4), 38 (2018).
- K. Usui, H. Kobayashi, Y. Fujita, E. Kubota, T. Hanazawa et al., J. Food Drug Anal. 27(3),
786–792 (2019). - K. Usui, E. Minami, Y. Fujita, E. Kubota, H. Kobayashi et al., J. Pharm. Biomed. Anal. 174, 175–181 (2019).
- European Union commission decision 2002/657/ec. Off. J. Eur. Communities 221, 8–36 (2002).
- S. Dulaurent, S. El Balkhi, L. Poncelet, J.-M. Gaulier, P. Marquet, and F. Saint-Marcoux, Anal. Bioanal. Chem. 408(5), 1467–1474 (2016).
Pauline Griffeuille is an intern in pharmacy. In November 2020, she started a PhD thesis in analytical chemistry. The main objective of her work is to develop analytical tools based on a PESI-MS/MS approach for applications in forensic, clinical, and occupational toxicology. This thesis is supervised by Sylvain Dulaurent and Franck Saint-Marcoux.
Sylvain Dulaurent is an engineer in analytical chemistry and obtained a Ph.D. in 2010. He is currently the analytical scientist responsible for the Clinical and Forensic Toxicology Unit of the Limoges University Hospital (France). He has developed and published numerous analytical solutions for the determination of pesticides residues, drugs, and illicit drugs. He is also a legal expert in forensic toxicology with daily activity dedicated to DUID.
Stephane Moreau obtained his diploma from INSA in fine chemistry and engineering with a specialization in chemical process engineering in 1994. He then started his professional career in laboratory equipment distribution before joining the brand new Shimadzu France subsidiary in 2002. Since then, Moreau has held various positions to develop the MS range of the business. Since September 2013, he has been product manager for the MS range with Shimadzu Europe.
Franck Saint-Marcoux works equally for the university and the hospital, where he is in charge of clinical toxicology, environmental toxicology, and forensic science for both. He is responsible for the Clinical and Forensic Toxicology Unit of the Limoges University Hospital (France). He is full professor of toxicology at the Faculty of Pharmacy of Limoges. He has received different scientific awards from international societies, and is author or co-author of over 80 papers in peer‑reviewed international journals.
Articles in this issue
over 4 years ago
Novel GC–MS Software Programs for Analyzing Seized Drug Samplesover 4 years ago
Controlling Retention Time Drift in Industrial Chromatographyover 4 years ago
FAST Updateover 4 years ago
SEP21 Previewover 4 years ago
HTC-17 Previewover 4 years ago
Octagon Therapeutics Receives Agilent Golden Ticketover 4 years ago
LCGC Europe and HTC-17 Launch HTC Innovation Award for 2022over 4 years ago
Shimadzu Receives Red Dot Awardover 4 years ago
Differentiating Frontotemporal Dementia Through MetabolomicsNewsletter
Join the global community of analytical scientists who trust LCGC for insights on the latest techniques, trends, and expert solutions in chromatography.