Articles by Christopher A. Pohl

The principal aim of this work was to provide a perspective with practical utility in streamlining the chromatographic method development in pharmaceutical industries based upon predicting the chromatographic retention times from molecular structures. Workflows were suggested with a focus on reversed-phase LC, IC, and HILIC as the three major techniques. Unlike HILIC, retention prediction in both reversed-phase LC and IC can benefit from the maturity of these techniques and the transparency of their retention mechanisms. In reversed-phase LC the solute coefficients in the hydrophobic subtraction model and in IC the a and b values in the linear solvent strength model can be the subject of modelling with their subsequent use in retention prediction. A workflow for HILIC can be based on the design of experiments approach, to account for all major contributors to the retention mechanism, and direct correlation of experimental retention times to the molecular descriptors.

This article describes a 2D matrix elimination ion chromatography method to analyse samples containing high levels of matrix ions.
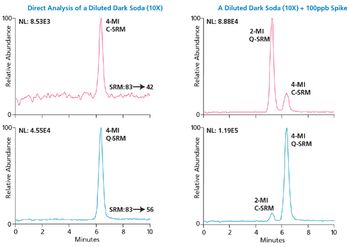
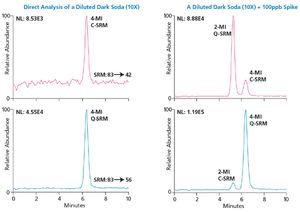
The chemicals 2- and 4-methylimidazole (2-MI and 4-MI) are by-products produced during the manufacturing of caramel coloring ingredients used to darken food products such as carbonated beverages and soy sauce.

Salt formation is a critical aspect in drug development and HPLC is an important tool for determination of pharmaceutical counterions.

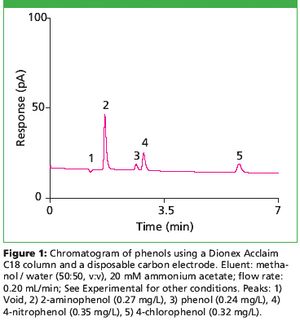
Phenols are frequently present in water because of their widespread use in commercial products and because they are by-products of processes in petrochemical, pulp and paper, plastic, and glue manufacturing industries (1,2). The concentration of phenolic compounds in the waste discharges can be as high as 20 mg/L (2); however, phenol-containing pesticides and wood preservatives may cause significant health hazards even at mg/L levels (1). Consequently, it is important to monitor phenols and substituted phenols in environmental and biological samples. Liquid chromatography with electrochemical detection is one of the widely used methods due to its high selectivity and sensitivity for phenolic compounds. However, glassy carbon working electrodes, used in the electrochemical detection of phenols, often require polishing (3). This time-consuming and often poorly reproducible polishing can be avoided with disposable carbon electrodes, which offer comparable or better analytical performance (4).
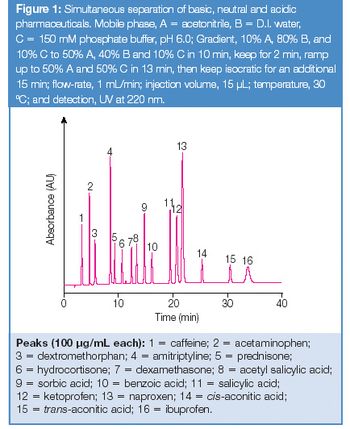
The separation and quantitation of drug substances and counterions are two important determinations in the pharmaceutical industry (1). Drug substances and counterions are determined by HPLC (often on reversed-phase columns) and by ion chromatography (IC), respectively. IC is the preferred method for selective and sensitive screening of both cationic and anionic pharmaceutical counterions (2). To increase the analysis throughput, it is desirable that for analysis of drug formulation both drug substance and counterions can be determined within a single run. Naproxen is a non-steroidal anti-inflammatory drug commonly used for treating moderate to severe pain, fever, inflammation, and stiffness. Naproxen sodium was approved by the U.S. Food and Drug Administration (FDA) as an over-the-counter drug. No reports were found describing one technique for the simultaneous determinations of both naproxen and the counterion (Na+).
The recent development of ultra-HPLC (UHPLC) has provided a great potential for high throughput analysis achieved using small (sub-2 μm) particle size columns at increased linear velocities. The advantage of UHPLC over conventional HPLC is increased throughput without sacrificing resolution. Despite their popularity, sub-2 μm particle columns impose practical difficulties, such as high backpressure (which often requires a UHPLC system) and susceptibility to column fouling. Thus difficulty can be overcome using 2.0 to 2.5 μm particles. This study describes an example (analysis of vanilla extract) of transferring a conventional LC method to a high-throughput method using newly developed Acclaim® RSLC 2-μm columns that are based on spherical, porous, high-purity silica particles (dp = 2 μm, pore size = 120 Å, surface area = 320 m2/g).
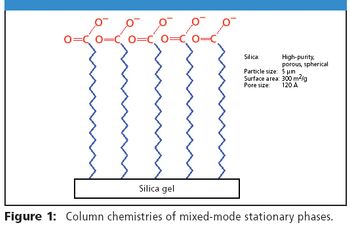
Mixed mode chromatography combines aspects of ion exchange chromatography and conventional reversed-phase (RP) chromatography. The combination of both hydrophobic and ion-exchange properties allows for independent control of retention for ionizable and neutral molecules.
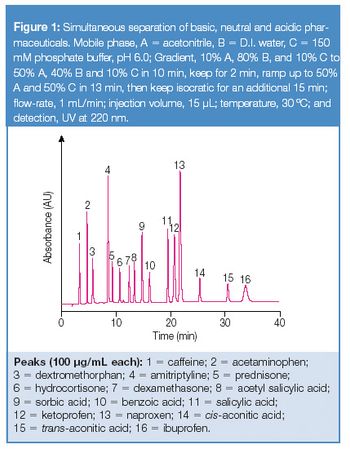
Xiaodong Liu and Christopher Pohl, Dionex Corporation