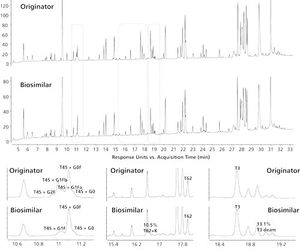
These are exciting times to be involved in monoclonal antibody (mAb) and biopharmaceutical analysis. Advances in instrumentation, column technology, and reagents are providing analysts with a new set of tools to broaden their understanding of the highly complex products they are studying. A good example is hydrophilic interaction chromatography (HILIC). While the technique has been used for more than 20 years to profile enzymatically released and fluorescently labelled N-glycans, the introduction of new columns (sub-2-µm and widepore) has paved the way to explore the technique further. Remarkable separations at all levels of analysis, including protein, peptide, and glycan levels, have been demonstrated. With data from the authors’ laboratories, the versatility of HILIC in mAb analysis will be demonstrated in this month’s “Biopharmaceutical Perspectives”.