Articles by Isabel Vandenheede

Monoclonal antibodies (mAbs) are being developed at an explosive rate and have attracted great interest from both smaller biotech firms and big pharmaceutical companies. Developing mAbs and next-generation antibody–drug conjugates (ADCs) is highly demanding in many ways. From an analytical perspective, handling mAbs and ADCs presents many new challenges. This article describes how size-exclusion chromatography (SEC) combined with high-resolution mass spectrometry (HRMS) can be applied to the detailed characterization of mAbs and ADCs.

The structural complexity of monoclonal antibodies (mAbs) challenges the capabilities of even the most advanced chromatography and mass spectrometry techniques. This study examines the use of micro-pillar array columns in combination with mass spectrometry for peptide mapping of both mAbs and antibody–drug conjugates (ADCs).

Monoclonal antibodies are becoming a core aspect of the pharmaceutical industry. Together with a huge therapeutic potential, these molecules come with a structural complexity that drives state-of-the-art chromatography and mass spectrometry (MS) to its limits. This article discusses the use of micro-pillar array columns in combination with mass spectrometry for peptide mapping of monoclonal antibodies (mAbs) and antibodyÐdrug conjugates (ADCs). Micro-pillar array columns are produced by a lithographic etching process creating a perfectly ordered separation bed on a silicon chip. As a result of the order existing in these columns, peak dispersion is minimized and highly efficient peptide maps are generated, providing enormous structural detail. Using examples from the author’s laboratory, the performance of these columns is illustrated.

These are exciting times to be involved in monoclonal antibody (mAb) and biopharmaceutical analysis. Advances in instrumentation, column technology, and reagents are providing analysts with a new set of tools to broaden their understanding of the highly complex products they are studying. A good example is hydrophilic interaction chromatography (HILIC). While the technique has been used for more than 20 years to profile enzymatically released and fluorescently labelled N-glycans, the introduction of new columns (sub-2-µm and widepore) has paved the way to explore the technique further. Remarkable separations at all levels of analysis, including protein, peptide, and glycan levels, have been demonstrated. With data from the authors’ laboratories, the versatility of HILIC in mAb analysis will be demonstrated in this month’s “Biopharmaceutical Perspectives”.

Two-dimensional liquid chromatography (2D-LC) has in recent years seen an enormous evolution, and with the introduction of commercial instrumentation, the technique is no longer considered a specialist tool. One of the fields where 2D-LC is being widely adopted is in the analysis of biopharmaceuticals, including monoclonal antibodies (mAbs) and antibody–drug conjugates (ADCs). These molecules come with a structural complexity that drives state-of-the-art chromatography and mass spectrometry (MS) to its limits. Using practical examples from the authors’ laboratory complemented with background literature, the possibilities of on-line 2D-LC for the characterization of mAbs and ADCs are presented and discussed.

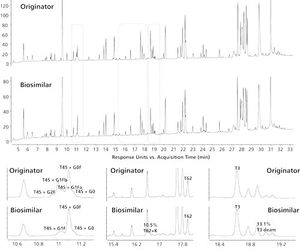
This article presents a selection of state-of-the-art analytical tools for mAb characterization and comparability assessment.

With the top-selling mAbs evolving out of patent there has been a growing interest in the development of biosimilars. In demonstrating comparability to the originator product, biosimilar developers are confronted with an enormous analytical challenge. This article presents a selection of state-of-the-art analytical tools for mAb characterisation and comparability assessment.

This article highlights some selected examples of the power of liquid chromatography combined with mass spectrometry (LC–MS) in the development of protein biopharmaceuticals.