LCGC North America
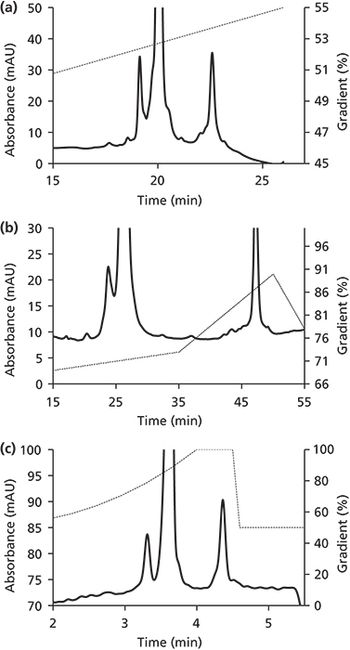
A systematic approach for formation of sigmoidal gradient for analysis of biopharmaceutical proteins has been reviewed and discussed together with two case studies. The first application involves HPLC separation of the various product related variants of a microbial expressed biotherapeutic, granulocyte colony stimulating factor (GCSF). The second case study involves separation of the charge heterogeneity related variants for a monoclonal antibody (mAb) biotherapeutic product. In all cases, it is observed that the use of sigmoidal gradient successfully reduces the analysis time significantly (from 70 minutes to 15 minutes for GCSF and from 40 minutes to 4 minutes for the mAb) while retaining the selectivity and the resolution.