- LCGC Europe-11-01-2017
- Volume 30
- Issue 11
Recent Developments of Comprehensive Two‑Dimensional Liquid Chromatography Coupled to Mass Spectrometry in Food Analysis
Food products are very complex mixtures containing organic and inorganic nutrients, and also xenobiotic substances that can derive from technological processes, agrochemical treatments, or packaging materials, including residues of pesticides, drugs, and toxins.
Food products are very complex mixtures containing organic and inorganic nutrients, and also xenobiotic substances that can derive from technological processes, agrochemical treatments, or packaging materials, including residues of pesticides, drugs, and toxins.
Food chemistry involves the assessment of food quality and authenticity, as well as the determination of molecules with a possible beneficial or toxic effect on human health (1). Analytical methods should be capable of determining the main components of food samples, and should also be selective and sensitive enough to determine minor components.
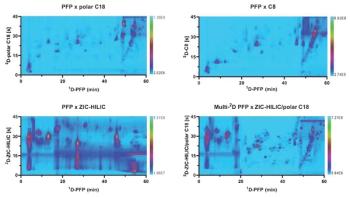
Liquid chromatography (LC) is characterized by a plethora of different separation mechanisms, namely normal phase, reversed phase, size-exclusion chromatography (SEC), ion exchange (IEX), affinity chromatography (AC), and hydrophilic interaction liquid chromatography (HILIC), which might be useful for tuning a higher number of potentially “orthogonal” combinations. The sample components are spread out according to two different retention patterns, over a range as broad as possible in respect of retention factor variation (2–5).
From its first application in 1978 comprehensive twoâdimensional LC (LC×LC) has been constantly investigated as a valuable tool to achieve much deeper insight into the true qualitative and quantitative composition of real-world food samples (6–7). In most cases, the outstanding selectivity and sensitivity of LC×LC methods combined with mass spectrometry (MS) detection made trace (ppb level), and ultratrace (ppt level and lower) analysis feasible, and reduced the need for tedious sample preparation processes.
Remarkable achievements in the field can be related to novel interface techniques, stationary phases, and mass analyzers.
The main developments in terms of interface techniques concerned the use of (a) actively modulated LC×LC (LC/a×m/LC) aiming to overcome one of the limitations of contemporary LC×LC arising from the combination of diverse first dimension (1D) and second dimension (2D) column diameters (8); (b) a “selective” LC×LC system (sLC×LC) aiming to break the long-standing link between the timescales of the 1D and 2D separations through novel implementation of existing valve technology (9); and (c) novel longitudinal on-column thermal modulation (LOCTM) with potential use in LC×LC systems involving a systematic optimization in terms of heat transfer, analyte retention, and migration velocity at a range of temperatures (10).
At the HPLC 2017 symposium in June (Prague, Czech Republic), a membrane evaporative interface was presented for the coupling of two independent dimensions (11). The device is placed before the switching valve to eliminate the 1D mobile phase, by evaporation through a hydrophobic membrane for improved LC×LC quantification capabilities and separation performance.
The most recent significant development in the practice of LC×LC has been the introduction of 2D small particle stationary phases with the aim of replacing the monolithic type that have been used previously for the same purpose. Notably in 2007, superficially porous stationary phases were developed to deliver “hyper-fast” chromatographic separations. Sub-2-μm technology was introduced later and was a milestone in LC×ultrahigh-pressure LC (UHPLC) applications, offering ultrafast and high-resolution 2D separations.
MS analyzers have evolved over the past ten years. In fact, quadrupole mass spectrometry (single stage [q-MS] or triple stages [QqQ-MS]); ion trap mass spectrometry (IT-MS), and time-of-flight mass spectrometry (TOF-MS) analyzers and hybrid MS detectors, such as Q-(or QqQ)âIT, QTOF, or IT-TOF, have been widely applied to attain a very significant gain in sensitivity, resolution, or speed for food applications.
It is reasonable to expect that the development of newly designed stationary phases, for example, capillary columns at nano-flow rate gradients, commercial instruments with reduced dead volumes, and high pressure valves will undoubtedly enhance the performance of LC×LC methods. Also, the development of more sophisticated and user-friendly software, allowing reliable and quick integration of 2D peaks, could be a valid tool for a widespread routine use of MS data for quantitative analysis. Outside of food analysis, a research field expected to rise in the next 5–10 years is in pharmaceutical analysis with a particular focus on the use of chiral stationary phases as the 2D in LC×UHPLC separations (12). Such an approach could be advantageously applied to the separation and analysis of complex mixtures of closely related pharmaceuticals and synthetic intermediates, constitutional isomers, stereoisomers, and organohalogenated species.
References
- W.R. Alley, B.F. Mann, and M.V. Novotny, Chem. Rev. 113, 2668–2732 (2013).
- Z. Liu, D.G. Patterson, and M.L. Lee, Anal. Chem.67, 3840–3485 (1995).
- P.J. Slonecker, X. Li, T.H Ridgway, and J.G. Dorsay, Anal. Chem. 68, 682–689 (1996). H. Gu, Y. Huang, and P.W. Carr, J. Chromatogr. A1218, 64–73 (2011).
- D.R. Stoll, X. Li, X. Wang, P.W. Carr, S.E.G. Porter, and S.C. Rutan, J. Chromatogr. 168, 3–43 (2007).
- F. Cacciola, P. Donato, D. Sciarrone, P. Dugo, and L. Mondello, Anal. Chem.89, 414–429 (2017).
- A.F.G. Gargano, M. Duffin, P. Navarro, and P.J. Schoenmakers, Anal. Chem.88, 1785–1793 (2016).
- S.R. Groskreutz, M.M. Swenson, L.B. Secor, and D.R. Stoll, J. Chromatogr. A1228, 31–40 (2012).
- M.E. Creese, M.J. Creese, J.P. Foley, H.J. Cortes, E.F. Hilder, R.A. Shellie, and M.C. Breadmore. Anal. Chem. 89, 1123–1130 (2017).
- E. Fornells, B. Barnett, M. Bailey, R.A. Shellie, E.F. Hilder, and M.C. Breadmore, paper presented at HPLC 2017, Prague, Czech Republic, 18–22 June 2017.
- C.L. Barhate, E.L. Regalado, N.D. Contrella, J. Lee, J. Jo, A.A. Makarov, D.W. Armstrong,and C.J. Welch. Anal. Chem.89, 3545–3553 (2017).
Francesco Cacciola is an associate professor of food chemistry in the Department of Biomedical and Dental Sciences and Morphofunctional Imaging at the University of Messina, in Messina, Italy.
Articles in this issue
about 8 years ago
Hitting Thirtyabout 8 years ago
LC Column Technology: The State of the Artabout 8 years ago
GC: The State of the Artabout 8 years ago
LC Instrumentation: The State of the Artabout 8 years ago
Sample Preparation: The State of the Artabout 8 years ago
The Evolution of 3D Printingabout 8 years ago
Capillary Electrophoresis: The Past, Present, and Futureabout 8 years ago
The Past, Present, and Future of Multidimensional Gas ChromatographyNewsletter
Join the global community of analytical scientists who trust LCGC for insights on the latest techniques, trends, and expert solutions in chromatography.




