The benefits of a robustness assessment for the analysis of a NIST mAb using a wide pore C4 LC column are described.

The benefits of a robustness assessment for the analysis of a NIST mAb using a wide pore C4 LC column are described.
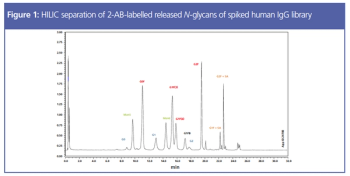
Post-translational modifications are potential critical quality attributes (pCQAs) routinely assessed in biotherapeutic development. Glycosylation is one of the most important attributes to assess because it affects protein function as well as antigen receptor binding. N-glycosylation of asparagine residues is the most common pCQA assessed during monoclonal antibody (mAb) therapeutic development. There are a few protocols to assess and quantitate N-glycans, but the most common approach is through an enzymatic release and labelling procedure, followed by separation and detection. This article demonstrates the method development considerations for sample preparation and chromatographic analysis of N-glycans of therapeutic mAbs.

This article describes the importance of magnetic bead and chromatographic media selection for hybrid LBA/LC–MS/MS workflows.

Within the broad scope of analytical techniques required to characterize a protein, chromatographic methods have shifted towards high-flow analyses that can drop development time significantly. However, fast analytical methods for charge heterogeneity have lagged in development because current column technologies are ultrahigh-pressure liquid chromatography (UHPLC)-incompatible. This article will demonstrate the development of a high-flow method for charge variant analysis made possible through a bioinert titanium column flow path.
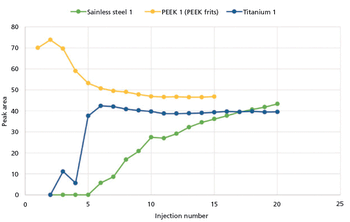
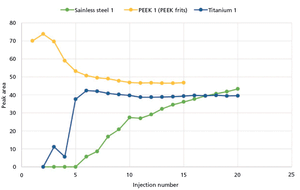
In this study, we compare the performance of plastic and metal materials in UHPLC columns designed for the analysis of biological molecules. We evaluate the performance of these materials in terms of inertness, column chromatographic performance, and reproducibility.

Antibody–drug conjugates (ADC) are an emerging pharmaceutical technology, with the potential of being the true “magic bullet”. However, these inherently complex biomolecules have unique analytical challenges. One is the determination of the drug-to-antibody ratio (DAR). The average DAR and the drug distribution need to be monitored and could determine the success of an ADC. This article discusses the chromatographic methods used to determine DAR, including hydrophobic interaction chromatography (HIC), reversed phase, and liquid chromatography–mass spectrometry (LC–MS).

Gel filtration chromatography (GFC) is the most widely used method for quantitating protein aggregates in therapeutic drugs. It is a simple method, but prone to error as a result of poor method development and column selection. GFC columns tend to non-specifically adsorb large proteins and aggregates resulting in poor quantitation of “true” aggregate amount. Sample “priming” and mobile phase optimization can help reduce such irregularities. Simple method development rules using new column technologies are presented that demonstrate improved accuracy for these methods.

The paramount problem in performing absorption, distribution, metabolism, and excretion (ADME)/pharmacokinetic studies of therapeutic oligonucleotides revolves around inefficient and labor-intensive sample preparation. These traditional methods require multiple steps - liquid–liquid extraction (LLE) and solid-phase extraction (SPE) - to extract therapeutic oligonucleotides from serum and plasma for liquid chromatography–mass spectrometry (LC–MS) analysis and thus are not practical for clinical studies with large numbers of samples. Furthermore, these methods tend to yield low recoveries and poor reproducibility. This article presents a revolutionary new method for performing sample cleanup of therapeutic oligonucleotides from serum and plasma. The method extracts many types of therapeutic oligonucleotides from biological matrices in a rapid four-step SPE protocol that eliminates the need for LLE and can be automated for large sample sets. In the testing presented, different..

Published: July 1st 2009 | Updated:

Published: December 1st 2015 | Updated:

Published: June 20th 2017 | Updated:
Published: June 1st 2018 | Updated:

Published: November 12th 2018 | Updated:

Published: February 5th 2020 | Updated: