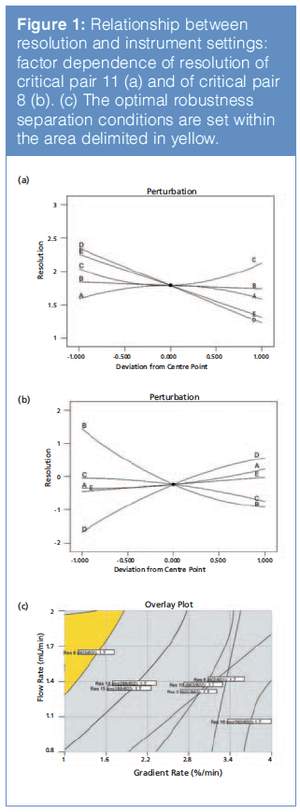
In this article, we review some key considerations for chromatographic technique selection and method development across the full drug process—from early-stage active pharmaceutical ingredient (API) synthesis to routine commercial release activities.