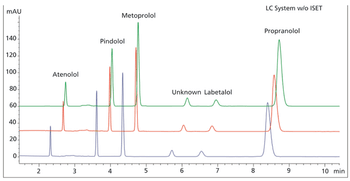
In method transfer, carefully aligning the modules of the two systems is essential.

In method transfer, carefully aligning the modules of the two systems is essential.

Scientists executing a method transfer often do not have access to the originating system. Thus, alternative approaches to matching chromatographic results must be considered.
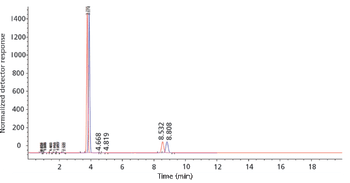
In the first of a three part series, we discuss challenges associated with successful transfer of chromatographic methods and how to best circumvent and remove ambiguities and contradictions

A discussion of how UHPLC is used to conduct intact protein–antibody analysis and glycoprofiling to characterize biopharmaceutical drugs

A description of the use of UHPLC in the biopharmaceutical industry to characterize drug substances through peptide mapping and amino acid analysis

UltraPerformance LC (UPLC) has been widely accepted by chromatographers because of improvements over HPLC in the sensitivity, resolution and speed of separations. As scientists begin to use this technology for impurity and metabolite profiling, they will need to transfer the methods to preparative LC to isolate and purify their compounds for further research. Therefore, it is necessary to systematically transfer UPLC assays not only to HPLC, but, more importantly, to preparative chromatography. In this application, we provide information on how to scale a UPLC impurity/degradant separation to a preparative LC separation.


Published: December 1st 2011 | Updated:

Published: September 1st 2011 | Updated:

Published: June 1st 2007 | Updated:

Published: June 6th 2005 | Updated:
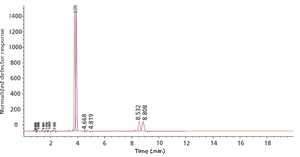
Published: September 1st 2018 | Updated:
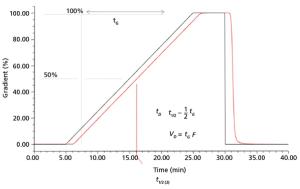
Published: November 1st 2018 | Updated: