
Oligonucleotides are in the center of many recent research projects including genetic testing, polymerase chain reactions (PCR), forensics, and DNA/RNA silencing.

Oligonucleotides are in the center of many recent research projects including genetic testing, polymerase chain reactions (PCR), forensics, and DNA/RNA silencing.

Anthrolysin (ALO) is a pore forming cholesterol-dependent cytolysin (CDC) secreted by Bacillus antracis.

Proteins of the Bcl-2 family are molecular transducers sensitive to internal and external apoptotic signals that play a key role in the regulation of apoptosis.

Carbon-clad zirconia phases offer dramatically different chromatographic selectivity for structurally similar compounds when compared to traditional silica-based bonded phases.

Deoxynivalenol is a common mycotoxin found in agricultural grain crops and final consumer processed products.

This study illustrates the successful combination of fast GC and tandem mass spectrometry to determine 360 pesticides spiked in a QuEChERS apple extract.

Pharmaceutical and personal care products (PPCPs) are products used for personal health or cosmetic reasons.
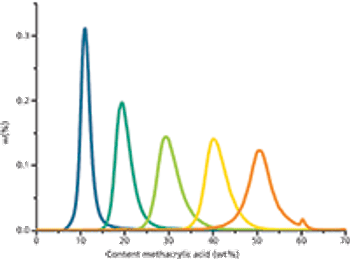
Statistic copolymers of methyl methacrylate (MMA) and methacrylic acid (MAA) are widely used in pharmaceutical applications.

The Agilent 1200 Infinity Series HDR-DAD Impurity Analyzer System covers a wide linear range typically up to 6000 mAU and enables the analysis of impurities typically down to 0.2 mAU.

Methadone is a commonly prescribed pain management type drug. As such, patients are monitored on a routine basis.

Monoclonal antibodies (mAbs) form a major class of biologics and recently biosimilars and biobetters are being added to the growing inventory of therapeutics.

Optimizing separation of ionizable compounds in order to find robust conditions has become an important part of method development in liquid chromatography.

Synthetic elastomers have replaced natural rubber to an astonishing degree, and account for more than 70% of the rubber used today.

Two species of spearmint oil (Mentha spicata and Mentha gracilis) are cultivated in the United States.
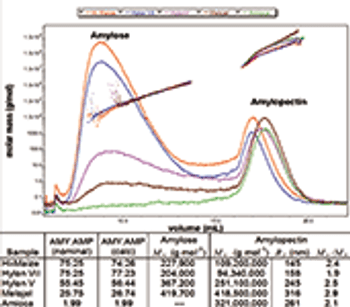
Starch is used for a variety of industrial and nutritional purposes. Its functional properties are influenced by the ratio and molar masses of its macromolecular constituents, which vary with source, crop year, and climate.

The unique surface chemistry of the FLARE diamond core-shell column combines ionic and hydrophobic separation mechanisms to effectively retain a variety of chemical species in a single run.




Wyatt Technology Corporation

The unique surface chemistry of the Flare diamond core-shell column combines ionic and hydrophobic separation mechanisms to effectively retain a variety of chemical species in a single run.

Modified starches are important materials used in many applications including foodstuffs.

High resolution time-of-flight mass spectrometry was used for comprehensive profiling of tobacco plant extracts.

Poly(lactic-co-glycolic acid) (PLGA) is a copolymer based on glycolic acid and lactic acid. The two monomer units are linked together by ester linkages and form linear polyester chains.
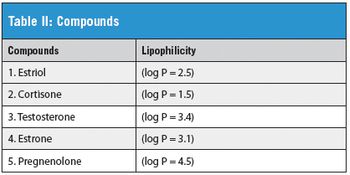
Steroids represent a chemically distinct class of hormones with wide-ranging biological functions. Synthetic derivatives of endogenous steroid prototypes are used medically in birth control and in the treatment of asthma, arthritis, inflammation, and osteoporosis.

The dioxin family consists of 210 compounds, of which 17 contain the 2,3,7,8 pattern of chlorination.

Natural and synthetic phenolic compounds have been used in a wide range of applications, from antiseptics and fungicides to food additives.

Pharmaceutical and personal care products (PPCPs) are products used for personal health or cosmetic reasons.

In mass spectrometry instrumentation, the sensitivity and resolution of the instrument is often limited by the detector.

Monoclonal antibodies (mAbs) form a major class of biologics and recently biosimilars and biobetters are being added to the growing inventory of therapeutics.