Implementing a peptide screening SPE plate determines the appropriate choice for a wide range of chemically diverse peptides.

Implementing a peptide screening SPE plate determines the appropriate choice for a wide range of chemically diverse peptides.

The optimized dual solid phase extraction (SPE) method was developed to significantly reduce the surfactant background from formulated drug products.

A HILIC solid phase extraction (SPE) and LC method that results in sensitive, effective, and a time savings for the clean-up of N-Glycans.

The development of a selective and rugged LC–MS/MS method for the quantitation of Amlodipine enantiomers in human plasma.

GenX, along with 24 other PFASs, are analyzed simultaneously to achieve sub-ppt reporting limits.

A reliable and efficient method was developed to determine the concentration sterols in olive oil by GC-FID.

The development of a selective and rugged LC–MS/MS method for the quantitation of Amlodipine enantiomers in human plasma.

SPE is used in combination with LC–MS/MS to analyze a panel of opiates from a urine sample.

Mycotoxin in grain are extracted using SPE to removes interferences followed by a rapid LC–MS/MS method.

Mycotoxin in grain are extracted using SPE to removes interferences followed by a rapid LC–MS/MS method.

GenX, along with 24 other PFASs, are analyzed simultaneously to achieve sub-ppt reporting limits.

A fast analytical method including both sample preparation and LC–ƒMS/MS analysis of ibuprofen in human plasma.

SPE is used in combination with LC–MS/MS to analyze a panel of opiates from a urine sample.

Optimize the LC–MS/MS workflow by using a SPE solution that provides in-well urine hydrolysis to save time.

Optimize the LC–MS/MS workflow by using a SPE solution that provides in-well urine hydrolysis to save time.

A reliable and efficient method was developed to determine the concentration sterols in olive oil by GC-FID.

The European Pharmacopoeia (Ph. Eur.) defi nes requirements for the qualitative and quantitative composition of medicines, as well as the tests to be carried out on medicines and on substances and materials used in their production.

For your highly sensitive UHPLC–MS analyses, how can you reduce noise and additional signals to a minimum?

Here we applied our newly developed UHPLC C18 and C30 columns to oligonucleotide analysis.

In this application, a simple method to simultaneously analyze various water-soluble vitamins was developed.
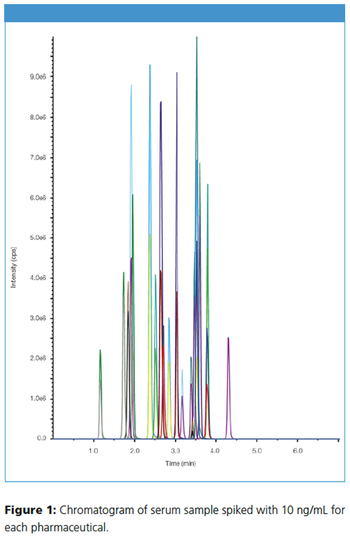
This application note describes the determination of pharmaceuticals from serum using solid-phase extraction (SPE) with the hydrophilic-lipophilic balanced SPE phase CHROMABOND® HLB for analyte enrichment and for sample cleanup. The eluates from SPE are finally analyzed by HPLC–MS/MS on a NUCLEOSHELL® PFP core–shell phase.

The amounts of high- and low-molecular-weight impurities are critical quality attributes for a therapeutic protein. Size-exclusion chromatography (SEC), the standard technology for analyzing aggregation and fragmentation, was applied to elucidate the molecular similarity between an Adalimumab biosimilar antibody and the corresponding innovator product Humira.

This application note describes the determination of pharmaceuticals from serum using solid-phase extraction (SPE) with the hydrophilic-lipophilic balanced SPE phase CHROMABOND® HLB for analyte enrichment and for sample cleanup. The eluates from SPE are finally analyzed by HPLC–MS/MS on a NUCLEOSHELL® PFP core–shell phase

Drug-antibody ratio (DAR) is a critical attribute of antibody–drug conjugates (ADCs). DAR may often be determined quickly and effectively by combining SEC with three online detectors: MALS, UV, and dRI.