Application Notes: Pharmaceuticals


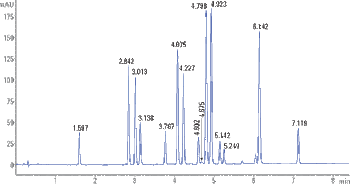
Pain management LC analyses can be difficult to optimize due to the limited selectivity of C18 and phenyl-hexyl phases. In contrast, the selectivity of Raptor Biphenyl superficially porous particle (SPP) LC columns provides complete resolution of isobaric pain medications with a total cycle time of 5 min.




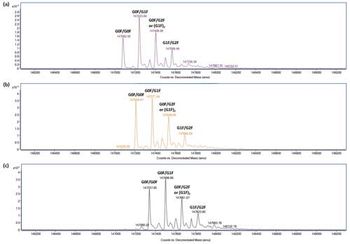
This application note demonstrates the analysis of charge variants of Rituximab (both innovator and biosimilar molecules) using multiple heart-cutting two-dimensional liquid chromatography (2D-LC) with mass spectrometry (MS) detection.


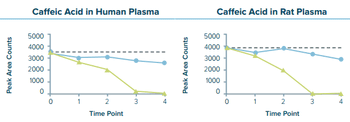
The ex vivo lability of some drug compounds and molecules in bio-analytical assays can add complexity and uncertainty to results and can pose significant challenges in drug development efforts. This can be especially true for compounds that are subjected to long-term cryogenic storage and that may need to be tested multiple times (for example, samples collected for clinical trials). Frozen aliquotting technology may offer a simple solution for stabilizing target compounds in frozen biological specimens, helping to streamline bio-analytical assay development and execution. The CXT 750 Frozen Sample Aliquotter, an automated instrument capable of generating quantitative aliquots of frozen plasma without thawing the sample, was evaluated at GlakoSmithKline for integration into biaonalytical workflows and the stabilization of labile compounds in EDTA plasma.

Shiseido succeeded in development of CAPCELL CORE ADME S2.7, a novel stationary phase with an adamantyl group having a cage structure. This is characterized as alkyl group; therefore, the packed column is still easy to use just as a reversed-phase column.

The peak resolution profile remained nearly unchanged. In particular, the resolution profile of thyroglobulin was similar to the resolution that was obtained by the 4.6 mm ID × 30 cm column. This suggests that the separation of high order molecular weight species, such as aggregates from mAb, can be easily achieved using this 4.6 mm ID × 15 cm column. Ten consecutive runs yielded excellent reproducibility. In fact, peaks from 10 consecutive injections were nicely overlaid.

TSKgel UP-SW3000 columns are 2 µm SEC columns designed for the analysis of monoclonal antibodies and other biopharma products. Higher resolution can be achieved for the separation of antibody monomers, dimers, and higher order aggregates with a TSKgel UP-SW3000 column compared to a competitor UHPLC column. The TSKgel UP-SW3000 column provided excellent reproducibility for the peak parameters of retention time, asymmetry, and column efficiency. As demonstrated by the %RSD values, injection-to-injection reproducibility was superior to the competitor column.




The ex vivo lability of some drug compounds and molecules in bio-analytical assays can add complexity and uncertainty to results and can pose significant challenges in drug development efforts.

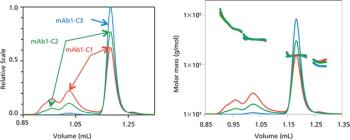
Therapeutic antibodies are enjoying high growth rates in the pharmaceutical market. A majority of the top bestselling global drug brands are monoclonal antibodies (mAbs).

In his article, published in 2000, David V. McCalley found large increases in the efficiency for basic compounds at elevated temperature

Complete separation of critical pain management drug analytes from hydrophilic matrix components and isobaric interferences was achieved using the new Raptor™ SPP Biphenyl LC column in less than 5 min

Avermectins are a series of 16-membered macrocyclic lactone derivatives that are used extensively in animal and crop protection

enable authentication of Si-Wu-Tang, a separate comprehensive 2D-LC analysis of each herb contained in Si-Wu-Tang was performed.

Glycosylation is one of the most common forms of post-translational modification of proteins. The polysaccharide side chains (glycans) play critical roles in physiological and pathological reactions. Besides the interest in characterizing glycosylation pattern of proteins for structure/function analysis, the thorough characterization of glycosylation is also a major quality parameter in the production of biotherapeutics. Hydrophilic interaction liquid chromatography (HILIC) is a well-recognized technique that effectively separates and quantifies isolated glycans.

Heparin is a polydisperse, heterogeneous polysaccharide derived from animal tissue. Heparin has been used as an anticoagulant for over 60 years and one of the fundamental parameters for characterizing unfractionated heparin (UFH) is its molecular weight (MW) and MW distribution. The molecular weight of heparin ranges from 3 to 50 kDa, but, typically falls within the 10 to 20 kDa range for unfractionated heparins used in medical applications. In light of heparin contamination issues in 2008, USP has proposed to include heparin molecular weight determination for the stage 3 heparin monograph revisions.

Most proteins, particularly soluble and membrane-bound proteins expressed in the endoplasmic reticulum, are glycosylated. The extent and result of glycosylation varies.

Organochlorine (OC) pesticides are hydrocarbons with multiple chlorine substitutions primarily used as insecticides. OC pesticides do not break down easily as the chlorine-carbon bonds are very strong.

Supercritical fluid chromatography (SFC) is an evolutionary technology and a powerful tool for enantiomer separation when used in combination with chiral stationary phases (CSPs).
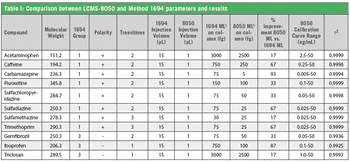
This method increases sensitivity and reduces data acquisition time for a subset of pharmaceuticals and personal care products from the original EPA Method 1694 specifications.

