
A review of advances in mRNA analysis using LC and LC–MS.

A review of advances in mRNA analysis using LC and LC–MS.

Gel filtration chromatography (GFC) is the most widely used method for quantitating protein aggregates in therapeutic drugs. It is a simple method, but prone to error as a result of poor method development and column selection. GFC columns tend to non-specifically adsorb large proteins and aggregates resulting in poor quantitation of “true” aggregate amount. Sample “priming” and mobile phase optimization can help reduce such irregularities. Simple method development rules using new column technologies are presented that demonstrate improved accuracy for these methods.

The global demand for fish as a natural source of fresh animal protein, essential fats, minerals, and vitamins continues to rise with the human population.
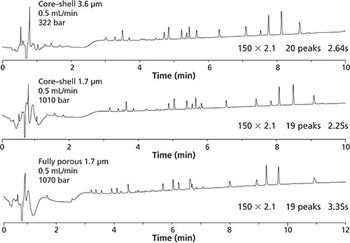
Ultrahigh-pressure liquid chromatography (UHPLC) columns are used in peptide mapping to improve the resolution of highly complex peptide mixtures. It is commonly assumed that small particle columns increase performance, but this is not always the case. This study presents a comparison of peak count, column length, and resolution between core–shell and fully porous UHPLC columns.

Although gel permeation chromatography (GPC) is not as widely used as other chromatography methods, it continues to be a useful technique for analyzing the size and solution characteristics of organic polymers. In this study, GPC was applied to analyze different polar polymers that are commonly used in drug excipients. As a result of their polarity, some specialized operating conditions were required. Optimizing the separation brought to light many of the common parameters involved in optimizing GPC separations, and those are discussed in detail here.

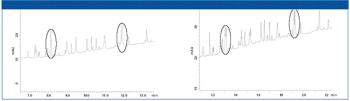
A new 3.6 µm 100 Å HPLC–UHPLC column (Aeris PEPTIDE) has been introduced that is specifically designed to improve separations of peptide and peptide mapping applications.

A new widepore core-shell HPLC–UHPLC column (aeris WIDEPORE) has been introduced that is specifically designed to improve protein separations.

A method is illustrated for the simultaneous analysis of ethyl glucuronide and ethyl sulfate in human urine samples.
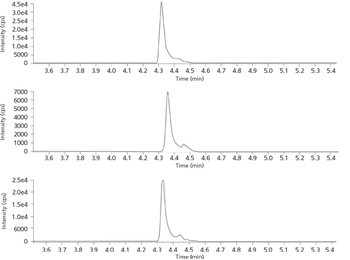
One of the biggest debates in clinical medicine currently revolves around understanding the proper levels of vitamin D in patients' plasma.

Increases in vitamin D testing frequency have required more rapid and cost-effective solutions for determining vitamin D levels in plasma. This LC–MS method was adapted for use with core-shell columns to achieve run times of less than 4 min.
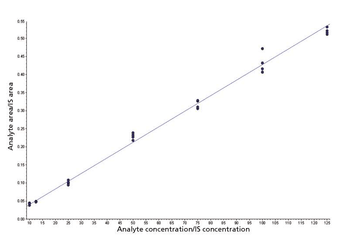
A high-throughput LC–MS method using core-shell UHPLC columns to screen for a panel of 11 drugs of abuse (expanded SAMHSA) was developed. The corresponding SPE method allowed the reproducible separation and quantitation of these 11 components in less than 2 min. This method demonstrates the power of new-generation HPLC media as well as some of the factors one must consider when developing such methods for LC–MS analysis.
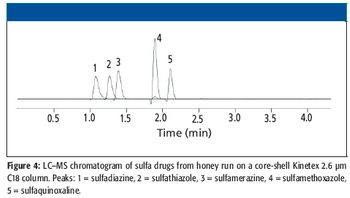
Adapting the use of "ultrahigh" performance chromatography for liquid chromatography–mass spectrometry (LC–MS) applications requires specific considerations in integrating the instrument platforms. Mobile phase options are limited to volatile buffers, and slow MS sampling rates can limit throughput advantages that such next-generation media offer. High-throughput LC–MS methods of different relevant pharmaceutical and environmental mixtures were developed using ultrahigh performance core-shell media. Such methods were developed using standard HPLC systems and back pressures, showing the ease and utility of using core-shell media for increasing throughput of LC–MS methods.

A critical part of research and quality control analysis of proteins involves verifying the primary and secondary structure of a protein. Peptide mapping is the main technique used to determine the structure of a protein as well as identify any post-translational modifications.

The paramount problem in performing absorption, distribution, metabolism, and excretion (ADME)/pharmacokinetic studies of therapeutic oligonucleotides revolves around inefficient and labor-intensive sample preparation. These traditional methods require multiple steps - liquid–liquid extraction (LLE) and solid-phase extraction (SPE) - to extract therapeutic oligonucleotides from serum and plasma for liquid chromatography–mass spectrometry (LC–MS) analysis and thus are not practical for clinical studies with large numbers of samples. Furthermore, these methods tend to yield low recoveries and poor reproducibility. This article presents a revolutionary new method for performing sample cleanup of therapeutic oligonucleotides from serum and plasma. The method extracts many types of therapeutic oligonucleotides from biological matrices in a rapid four-step SPE protocol that eliminates the need for LLE and can be automated for large sample sets. In the testing presented, different..

Additional studies were undertaken to better understand the chromatographic behavior of PEGylated proteins in an effort to improve purification and characterization techniques of such proteins. Proteins were PEGylated using larger (20 KDa and 40 KDa) PEGylation reagents that are commonly used in pharmaceutical drug development. Generated PEGylated proteins were separated from unmodified proteins using different reversed phase medias (Jupiter® C4 and Jupiter® C18). In these studies it was found that the Jupiter C18 media provided the best separation of PEGylated proteins from their unmodified counterparts. Such results further clarify good method starting points for developing analytical and preparative separations of PEGylated proteins.
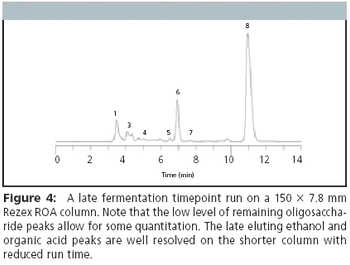
Political priorities as well as economic interests have fueled a dramatic growth in the biofuel industry, due much in part to research funding and tax incentives. Currently the world's ethanol production is estimated to be over 16 billion gallons a year. This number is expected to increase in the next few years, reaching an estimated yearly production of 20 billion gallons by 2012. Today, many ethanol producers add fermentors to expand their production capacity. In order to continue using existing HPLC equipment for the increased monitoring, increased analytical throughput is needed.

Published: January 15th 2024 | Updated:

Published: October 31st 2013 | Updated:

Published: July 1st 2011 | Updated:

Published: March 2nd 2012 | Updated:

Published: February 1st 2012 | Updated:

Published: February 1st 2012 | Updated: