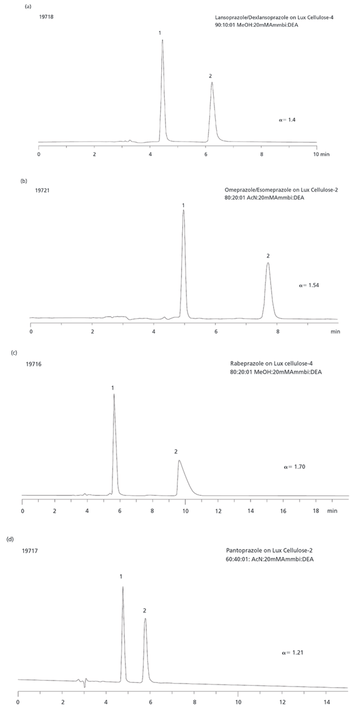
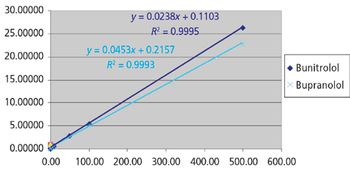
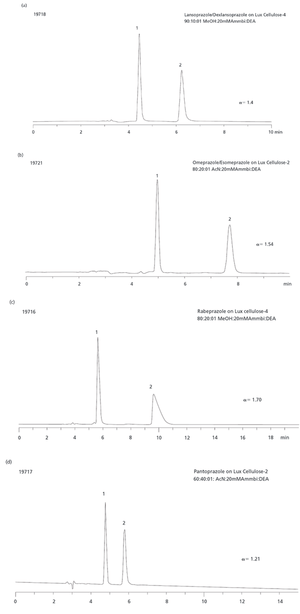
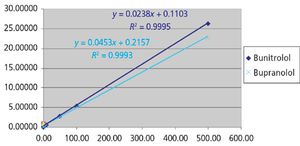
Beta-blockers are basic compounds that contain a secondary amino group in their structure. The amino substituents are typically an isopropyl group and a larger chain with a hydroxyl group in the beta position from the nitrogen atom (Table I). The simultaneous analysis of ?-blockers in biological samples is meaningful, and is made possible by the similarities in their structure. Gas chromatography (GC)–mass spectrometry (MS) has been the most used technique for their identification and quantification (4–6). However, most ?-blockers are nonvolatile and thus require derivatization via a cumbersome and time-consuming process before GC–MS analysis. In recent years, liquid chromatography (LC) coupled with mass spectrometric detection has evolved as the method of choice for drug analysis in the pharmaceutical, clinical, and forensic toxicology areas (4–8). In contrast to GC–MS, LC–MS-MS generally does not require derivatization and offers superior sensitivity. Moreover, due to the high specificity offered by LC–MS-MS, baseline chromatographic resolution often is not required, allowing for fast analysis in high-throughput environments.