
Thermo Fisher Scientific
Articles by Thermo Fisher Scientific



Professor Jesper Olsen sets a new world record for identifying the maximum number of unique peptides per minute. Time and Date: Friday, Sept. 29, 2017 | 11 am EDT | 8 am PDT | 4 pm BST | 5 pm CEST / On Demand available until Sept. 29, 2018 Register Free: http://www.chromatographyonline.com/lcgc_w/benchmarking



High resolution mass spectrometry with nano-LC is used for protein identification and quantification in both top-down and bottom-up proteome analysis. Reliable instrumentation in combination with ultrapure mobile phases is essential for data integrity. Premixed 80% acetonitrile with 0.1% formic acid (LS122-500) and water with 0.1% formic acid (LS118-500) were designed to produce a consistent chromatographic performance using this instrument system. In this study, these mobile phases were used extensively to evaluate several factors which can affect separation of protein digests such as peak retention, peak repeatability, and sample carryover. Our results demonstrated excellent chromatographic performance using Thermo Scientific EASY-nLC 1200 LC system and Thermo Scientific LTQLX ™ ion trap mass spectrometer with the specialized premixed mobile phases.

In recent years, food analysis has blossomed out from a singular focus on food safety to a studies of a wide array of questions such as the authentication of food origin and the nutritional aspects of natural food compounds. In this new e-book, we explore some current trends in food analysis and also provide some concrete advice for preparing food samples for analysis.

Food and beverage manufacturers who perform simple sugar analysis require methods that provide sensitive, selective, and direct determination of carbohydrates. Minimizing errors associated with sample preparation is of key importance, because samples often require large dilutions that can impact the downstream accuracy of results. In this summary, the benefits of using high-performance anion-exchange chromatography coupled with pulsed amperometric detection (HPAE-PAD), which does not require sample derivatization, will be discussed. We will also cover recent technological advances, including high pressure ion chromatography, which have enabled manufacturers to analyze more samples in less time.


Gas chromatography (GC) is a powerful technique for environmental analysis. The articles in this new e-book look at both the technique and its application, providing essential information about GC fundamentals as well as an example of how the technique can be used in new ways in environmental analysis.

In this Q&A, we will talk to Dr. Arthur Fitchett, Director of Sales Training at Thermo Fisher Scientific. We will discuss recent advances in ion chromatography technology, what they mean for different types of IC analyses, and how they can improve results in your laboratory.

Your current contract laboratory and instrument provider may be familiar and comfortable, but, maintaining the status quo doesn’t lead to greater rewards. In fact, it often involves putting up with less-than-desired results. Join us in this executive summary to hear how Ajinomoto Althea develops unsurpassed solutions for biopharma companies, and how a team at Thermo Fisher Scientific has challenged the status quo to deliver uncompromised UHPLC.

This podcast will focus on new developments involving our understanding of disinfection by products in drinking water. We will cover updates to toxicological compounds and advancements in new technologies to identify them.

Ion chromatography (IC) is a well-established technique for monitoring inorganic anions in environmental waters around the world. It is approved by the U.S. Environmental Protection Agency (U.S. EPA) for compliance monitoring of primary and secondary inorganic anions in drinking water and has been validated by European standards organizations, including the International Standards Organization (ISO). These agencies have published IC methods for the analysis of inorganic anions in drinking water, groundwater, and wastewater. These methods can be made faster and more cost-effective, however, by using the latest IC technologies, including high-pressure IC. In this summary, we will discuss regulatory compliance testing of municipal drinking water and wastewater, and how this testing can be made faster and more cost-efficient through the use of high-pressure-capable ion chromatography systems.

This summary will explore the latest developments in the analysis of contaminants of emerging concern in drinking water. We will explore the requirements and results from the EPA’s Unregulated Contaminant Monitoring Rule 3 program for Perfluorinated organics, and hormones. After a brief overview of the EPA analytical techniques, we’ll take a look at new technology and ask how it compares for both and if it can find compounds we didn’t even know were there.

Ion Chromatography Applications and Detection Methods from the Thermo Scientific portfolio of products for the detection of inorganic anions and cations, toxic contaminants, and disinfection byproducts.

Determination and quantification of water contamination by phenols using the Dionex AutoTrace 280 SPE instrument.

A method incorporating direct analysis in real time (DART) ionization and Thermo Scientific™ Orbitrap™ high-resolution mass spectrometry for rapid analysis and identification of contaminating substances in water.

Specifications and data highlighting the Thermo Scientific™ Dionex™ IonPac™ CS16-4μm and CS16-Fast-4μm cation-exchange columns for the determination of disparate concentration ratios of sodium and ammonium in diverse sample matrices.

Automated Solid-phase extraction (SPE) with GC-ECD for the determination of 15 nitrobenzene compounds in finished drinking water using the automated Thermo Scientific™ Dionex™ AutoTrace 280 Solid Phase-Extraction instrument in conjunction with the Thermo Scientific™ TRACE™ 1310 gas chromatograph.

Development of an ion chromatography (IC) method using suppressed conductivity detection for the determination of Cr(VI) in drinking water.

A method for the determination of pyrethroids in water at ultra-low-level concentrations of 0.02 and 0.10 ng/mL was developed using solid-phase extraction (SPE) for pre-concentration and subsequent analysis by GC with PTV injection.

Analysis of OCPs, PAHs and PCBs in environmental samples through a consolidated GC-MS/MS method using the Thermo Scientific™ TRACE™ 1310 GC and the TSQ™ 8000 triple quadrupole GC-MS/MS.

The introduction of UHPLC revolutionized bio/pharma analytical laboratories and advances in technology promise more for the future. In this ebook, experts explain recent advances in UHPLC, and the basics of glycan analysis. Sample preparation technologies that can increase sample throughput and improve data quality for peptide quantitation are explored. In addition, the characterization of intact antibodies using reversed-phase chromatography is detailed. Review UHPLC workflows for: • Glycan analysis • Peptides • Monoclonal antibodies

High pressures and columns with small particle sizes have been used in high-performance liquid chromatography (HPLC) for a long time, but not in ion chromatography (IC). But now, high pressures are coming to IC. Read this new e-book to learn about: • The advantages of high-pressure and small-particle-size columns in IC • How those advantages apply to real-world examples in the analysis of food, beverages, and environmental samples • The reasons why the move to high-pressures in IC has taken so long • The drivers for the trend toward higher-capacity IC columns . . . and more! Download the e-book today!
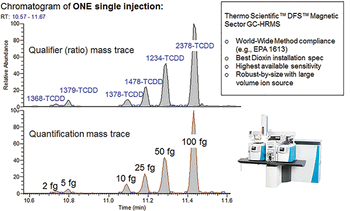
The standards are commercially available from Wellington Laboratories Inc. (Ontario, Canada): TF-TCDD-MXB.

Dr. Graeme McAlister of Thermo Fisher Scientific will discuss the advances in Orbitrap Tribrid Mass Spectrometry technology with the launch of the new Thermo ScientificTM Orbitrap FusionTM LumosTM TribridTM Mass Spectrometer. Graeme will also touch on how these advances have impacted and will continue to impact proteomics with respect to quantitation, accuracy, throughput, etc.

In this webinar, we will discuss the use of new, affordable HRMS instruments in quantitative clinical research analyses, demonstrating the sensitivity and selectivity of these instruments for this work. Examples of clinical research quantification work in HR full scan or MS-MS mode will be presented and compared with results from triple-quadrupole-MS. We will also discuss an example of Qual/Quan analysis in a study of the fate of an anti-cancer agent in humans, where over 40 metabolites were identified and quantified. We will also show metabolomics data underscoring the versatility of HRMS instruments.

The application of high resolution, accurate mass (HRAM) mass spectrometry coupled with multidimensional chromatographic separations, bioaffinity isolation techniques, and online sample preparation methods to detect, identify, and quantify emerging contaminants (including endocrine disruptors, pharmaceuticals, and surfactants) in wastewater and drinking water. The overarching goal of Lee’s research is to gain an increased understanding of how emerging contaminants are transported, transformed and induce deleterious effects within aquatic ecosystems.
