- July / August 2023
- Volume 36
- Issue 07
- Pages: 267–274
Polysorbate Analysis in Biopharmaceutical Applications—A Snapshot of the Analytical Toolbox

Polysorbates (PS) are added to biopharmaceutical formulations to stabilize proteins and monoclonal antibodies (mAbs) and to prevent aggregation, denaturation, and surface adsorption. Polysorbate analysis can be challenging because of their inherent complexity, polydispersity, and their presence in matrices containing high concentrations of proteins and other excipients such as sugars, amino acids, salts, and buffers. This instalment of “Biopharmaceutical Perspectives” provides an overview of different approaches for the qualitative and quantitative analysis of polysorbates, its impurities and degradants in raw material, intermediate dilutions, drug substance, and drug products. Various gas (GC) and liquid chromatography (LC) methods and accompanying detection techniques are briefly discussed and illustrated with results obtained at the authors’ laboratory.
Polysorbates (PS) are a class of nonionic surfactants mainly used in the food and pharmaceutical industry. In the biopharmaceutical field, polysorbate 20 (PS20) and 80 (PS80) are widely accepted and prepared at concentrations between 0.01–0.05%—well above the critical micelle concentration (1–4). These key excipients have low toxicity and feature stabilizing properties, thereby countering aggregation, denaturation, and surface adsorption of proteins and monoclonal antibodies (mAbs) in solution. As a result of their chemical nature, polysorbates are prone to degradation, which is influenced by several factors (1–8). Polysorbate degradation has emerged as a hot topic in the industry in recent years, predominantly because of fatty-acid mediated particle formation in PS-containing formulations, and has been identified as one of the main challenges during formulation development of protein biopharmaceuticals. Understanding the origin of degradation and the implementation of control and mitigation strategies have been widely discussed (3,4,6). Analyzing PS is therefore fundamental, and has resulted in the development of different methodologies that are becoming an essential part of the analytical arsenal for protein biopharmaceuticals.
Structural Heterogeneity of Polysorbates
Polysorbates are heterogeneous amphiphilic substances composed of a hydrophilic polyoxyethylene (POE) sugar head group and esterified fatty acids that make up the hydrophobic tail (Figure 1). In contrast to other small molecules that are defined by a specific structure, its chemical synthesis generates a complex mixture, with PS heterogeneity originating from the type of fatty acids (lauric, myristic, palmitic, stearic, oleic, linoleic, linolenic), esterification degree (0–4), sugar structure (sorbitan, isosorbide), and POE chain length (target ethylene oxide units equals 20), giving rise to well above a thousand possible species and a complex structure–function relationship (1,3,6). On top of this, the PS diversity among manufacturers and lot-to-lot variability adds to the complexity. The prevailing esterified fatty acid, lauric and oleic acid, respectively, differentiates PS20 and PS80. Remarkably, the target structure of the latter surfactants, that is, POE (x+y+w+z = 20) sorbitan monolaurate and POE (x+y+w+z = 20) sorbitan monooleate, is typically found to account for only 20% of the total PS species (1,3).
Stability of Polysorbates
While polysorbates are highly functional excipients, they are chemically and enzymatically labile and prone to degradation by oxidation and hydrolysis (Figure 2) (1–8). The oxidative pathway can be triggered by exposure of neat or formulated PS to air, light, and transition metals, and is influenced by the purity grade of the raw materials (for example, peroxide content). Hydrolysis can proceed in a purely chemical manner by heat or alkaline and acidic conditions, or by the presence of trace amounts of host-cell hydrolytic enzymes (esterases, lipases) co-purified with the therapeutic protein. The chemically induced hydrolysis pathway is uncommon in a biopharmaceutical context, while enzyme-mediated hydrolysis has been identified as the most challenging degradation in industry and typically occurs at high protein concentrations (1,4).
Different risks are associated with degradation through oxidation and hydrolysis. To a certain extent, oxidized PS is able to maintain its protein-stabilizing properties (7). Accumulation of degradation products, such as aldehydes, ketones, and peroxides, may nevertheless present a risk towards the chemical stability of the therapeutic protein, potentially impacting its efficacy (5). Peroxides can lead to oxidation of methionine, tryptophan, or histidine residues, while aldehydes react with primary amino groups (protein N-terminus or lysine) (1,7).
The hydrolytic degradation of PS can impact its functionality as a protein stabilizer (7). Furthermore, the main degradation products, free fatty acids (FFA), may form visible and subvisible particles when above their aqueous solubility threshold, drastically impacting drug product quality and shelf life (1,4,6). Particles in parenteral drug products carry risks associated with blood vessel occlusion during intravenous delivery and immunogenicity reactions during subcutaneous and intramuscular delivery.
The Analytical Toolbox
Polysorbate analysis within a biopharmaceutical context aims at determining PS identity, content (quantity), heterogeneity, variability, impurities, and degradants in the raw materials, intermediate dilutions, drug substance, or drug product, in stability or purposely stressed samples. Given the range of questions to be addressed and the structural complexity and solute diversity (from aldehydes to polyesters) encountered, a multitude of analytical methodologies has been developed, with some focusing on one analytical target (content determination) and others with a broader utility. A crucial part in each methodology is sample preparation, which typically consists of a protein removal step, enrichment, and derivatization of solutes to enable PS analysis.
The following sections will provide the reader with an overview of the chromatographic and nonchromatographic methodologies for PS analysis.
Nonchromatographic Methods
The fluorescence micelle assay (FMA) is well embedded in industry for the determination of PS content (1,3,9). The principle of the methodology is based on increasing the fluorescence emission of N-phenyl-1-naphthylamine (NPN) once it distributes to the hydrophobic core of a PS micelle. The assay, originally developed for critical micelle concentration determination (10), can be implemented in plate format or can be based on flow injection analysis (FIA) using high performance liquid chromatography (HPLC), with the dye present in the mobile phase and the interaction with PS taking place in a knitted reaction coil prior to fluorescence detection (Figure 3[a]). Run times are fast, but throughput is hampered by extensive equilibration runs (± 50) and the use of replicate sample and calibration runs to cope with the varying fluorescence signal. As NPN also interacts with other hydrophobic components, protein precipitation or dilution is usually required for protein concentrations > 50 mg/ mL. It should be noted that the assay has demonstrated stability indicating character that depends on the PS type and degradation mechanism.
Alternative nonchromatographic methods for PS quantification that do not rely on micelle formation, each with their own weaknesses and strengths towards sensitivity, matrix complexity, and stability‑indicating character, have also been described (1). These include spectroscopic assays based on the formation of coloured ferric or cobalt thiocyanate PS complexes or the differential emission of the amphiphilic fluorescent dye 1,1’-dioctadecyl-3,3,3’,3’-tetramethylindocarbocyanine perchlorate in polar and nonpolar environments (11). Colorimetric and fluorometric assays are used to determine the content and formation of peroxides at early stages of the oxidation cascade (1).
Chromatographic Methods
Liquid Chromatography:
HPLC methods have been widely applied for PS analysis because of their versatility, simplicity, richness in information, and general acceptance within the pharmaceutical industry. The absence of a chromophore restricts detection by UV, hence universal detectors, such as evaporative light scattering (ELSD) and charged aerosol (CAD) detectors, alongside mass spectrometry (MS), are implemented. All have similar requirements towards mobile phase volatility, offer good sensitivity in the encountered concentration range, and show compatibility with reversed‑phase LC (RPLC) and hydrophilic interaction liquid chromatography (HILIC).
The hydrophobic nature of PS renders this surfactant highly suitable for RPLC, and its excellent retentivity allows separation between different PS species to be achieved, facilitating characterization; alternatively, PS can be eluted into one single peak to simplify quantification (1,6,9,12–19). A protein removal step via solid-phase extraction (SPE) or precipitation is typically required. The latter can be circumvented to a certain extent using mixed-mode liquid chromatography (MMLC), which combines anion exchange and reversed-phase mechanisms, thereby enabling separation of nonionic PS from positively charged protein material and hydrophilic excipients (9,12,16). Applying a step gradient from an aqueous acidic mobile phase to one of higher organic content on a short mixed-mode cartridge results in the coelution of all PS species in the ELSD, as illustrated in Figure 3(b) for a PS80-containing mAb formulation. The implementation of a postcolumn switching valve to prevent ELSD contamination and increase method robustness hinders the detection of the mAb in the void volume. When using linear organic solvent gradients on state-of-the-art RPLC columns, PS species predominantly elute based on their esterification degree, thereby providing a fingerprint in comparability and stability studies as shown in Figure 3(d) for different PS80 raw materials (13–19). While degradation and batch inconsistencies can be monitored, PS quantification from such complex chromatographic profiles can be challenging, especially given the latter batch variability. To obtain a uniform CAD response under gradient conditions, the chromatograms in Figure 3(d) were obtained using a postcolumn inverse compensatory gradient. In the absence of this correction, the mass balance was disturbed with an overestimation of di-, tri-, and tetra-esters and an underestimation of nonesterified components. When combining MMLC and RPLC in an online heart‑cutting two‑dimensional (2D)-LC setup, PS can automatically be separated from the protein in the formulation and further resolved in the second chromatographic dimension (16). The resolution added by MS further extends the characterization capabilities and allows unambiguous identification of the species observed (13–19). Spectra collected over the RPLC peaks display mass envelopes, with characteristic 44 Da spacings corresponding to the ethylene oxide building blocks. PS is commonly transferred in the gas phase using electrospray ionization (ESI) operated in positive mode and—as a result of a lack of basic functional groups—detected as singly, doubly, and triply charged ammonium or sodium adducts depending on the mobile phase composition.
RPLC is valuable in measuring PS degradants or impurities such as FFAs or aldehydes and ketones— the latter entities following derivatization with 2,4-dinitrophenylhydrazine (DNPH) to facilitate detection by UV (1,20,21). The RPLC–MS analysis of FFAs in a PS20-containing mAb formulation is shown in Figure 3(c). Separation is based on carbon number, and degree of saturation and sensitivities down to 10 ng/mL can be achieved in negative ESI mode with a single quadrupole mass analyzer. FFA impurities and degradants can therefore be quantified in mAb formulations following protein precipitation. In the presented example, PS degradation is noticed in a stability sample because of the marginal presence of residual host-cell hydrolytic enzymes. To guarantee sensitive, precise, and accurate quantification, isotopically labelled fatty acid counterparts are typically used and precautions are taken to limit extrinsic fatty acid contamination. In the case presented, dedicated consumables and chemicals were used in addition to a reversed-phase isolator column installed prior to the autosampler to retain fatty acid contaminants, such as palmitic and stearic acid, originating from the mobile phase (21). While MS allows fatty acid measurement as such, early work reports on carboxylic acid derivatization using 1-pyrenyldiazomethane (PDAM) to render fatty acids fluorescent (1). Adequate sensitivity can be obtained at the expense of additional sample preparation.
HILIC exploits the polar regions of PS and offers complementary selectivity. The technology has been used less frequently in one-dimensional (1D) separations; however, combining it with RPLC in a comprehensive 2D-LC setup provides an unsurpassed fingerprint where small compositional changes originating from batch variability or degradation can easily be revealed (Figure 3[e]) (22). While HILIC resolves polysorbates based on the degree of ethoxylation and type of sugar (isosorbide, sorbitan), RPLC separation is predominantly based on esterification degree and carbon number.
Gas Chromatography:
Due to their molecular mass, polarity, and low volatility, polysorbates cannot be analyzed as such by gas chromatography (GC). Nevertheless, GC and GC–MS, in combination with various sample preparation methods, can provide highly relevant and complementary information in PS analysis.
PS quantification and characterization can be achieved by fatty acid analysis following transesterification. The raw material, intermediate dilution, drug substance, or drug product is treated with a derivatization reagent such as boron trifluoride (BF3) in methanol. The PS is hydrolyzed and fatty acids are converted to the fatty acid methyl esters (FAMEs), which, following liquid-liquid extraction using hexane, are analyzed by GC in combination with flame ionization detection (FID) or MS. A typical electron ionization (EI) total ion chromatogram (TIC) of a PS20-containing mAb formulation obtained on a polyethylene glycol (PEG)-based free fatty acid stationary phase (FFAP) is shown in Figure 4(a). Excellent separation according to carbon number and unsaturation is demonstrated and the relative fatty acid composition can be assessed and compared against PS batches, PS types, and stability samples. Changes in relative composition or loss of unsaturated fatty acids can indicate PS oxidative degradation. Determining the relative composition is performed by GC–FID because of the nearly constant detector response for fatty acids in the C10–C20 range. Using proper calibration, preferably with an identical PS batch, both GC–FID and GC–MS can be used for absolute PS quantification. Beyond FFAP columns, cyanopropyl stationary phases can be used for FAME analysis, offering the benefit of resolving cis/trans double bond isomers. The former PEG-based phase, however, has the advantage that it can also be used for FFA analysis, as demonstrated in Figure 4(b) for an aged PS20‑containing mAb formulation. Note that the above-described FAME analysis does not discriminate between free and esterified fatty acids in PS samples as all are converted to methyl esters. For FFA analysis by GC, sample preparation involves protein precipitation followed by liquid injection of the organic supernatants. To maximize sensitivity, the single quadrupole mass analyzer is operated in simultaneous scan and selected ion monitoring mode (SIM) transmitting the selective ions at m/z 60 (McLafferty rearrangement) and 73.
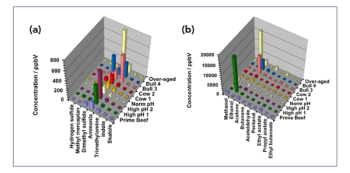
Various PS degradants resulting from oxidation are volatile and amenable to GC analysis in a targeted or untargeted manner. A typical example of targeted analysis is the determination of C1–C12 aldehydes and ketones. The sensitive and selective analysis of the latter species can be performed using solid-phase microextraction (SPME) in headspace (HS) mode combining extraction and in-fibre derivatization. Briefly, the procedure includes loading the SPME fibre with derivatization reagent by sampling the headspace of an aqueous solution of O-(pentafluorobenzyl)hydroxylamine (PFBHA), followed by SPME sampling in the headspace of the sample, thermal desorption of the derivatized aldehydes and ketones in the GC inlet, and GC–MS analysis. This procedure is typically automated using a dedicated autosampler. Figure 4(c) presents the results obtained for fresh and temperature-stressed PS20 raw material. The overlaid extracted ion chromatogram (EIC) at m/z 239, selective for saturated aldehydes, shows substantially increased levels of C4–C10 aldehydes due to stress.
Untargeted analysis allows a broader screening of degradants and impurities, and a highly sensitive approach is based on dynamic headspace sampling (DHS), where the headspace of a sample is purged with inert gas and the volatile organic compounds (VOCs) are exhaustively concentrated on an adsorbent trap (typically containing Tenax). The trap is subsequently thermally desorbed online using GC–MS analysis. Figure 4(d) shows the analytical ion chromatograms (AIC) obtained for unstressed and stressed PS20 material. Excellent separation of aldehydes, ketones, alcohols, and other VOCs was obtained using a polyethylene glycol GC column and untargeted detection facilitated by fast scanning time-of-flight (TOF)-MS and powerful deconvolution algorithms. An alternative complementary solventless extraction method that can be used is stir-bar sorptive extraction (SBSE) (5,23). SBSE allows the enrichment of apolar and semi-polar organic compounds from aqueous solutions, without the need for protein removal. As the method is solventless, it can be considered as a green analytical sample preparation, and solvent blank issues are minimized. While DHS has a bias towards the more volatile species, SBSE will preferentially sample less volatile compounds, including lactones.
Conclusions
With all eyes on this precious, yet moody, surfactant in the biopharmaceutical industry, a multitude of methodologies has been developed in recent years for PS analysis. This has substantially increased our understanding of PS structural diversity, variability, degradation, and structure/function relationship, and has led to various strategies to optimize its use in protein formulations. With dozens of creative analytical scientists being active in the field, more methods are expected to emerge in the coming years, further boosting our knowledge of this important subject.
Acknowledgement
The authors acknowledge Frank Steiner and Mauro de Pra (Thermo Fisher Scientific, Germering, Germany) for kindly providing the PS80 RPLC-CAD chromatograms and all the analytical Tween geeks at RIC group for their behind the scenes contributions.
References
(1) Martos, A.; Koch, W.; Jiskoot, W.; et al. Trends on Analytical Characterization of Polysorbates and their Degradation Products in Biopharmaceutical Formulations. J. Pharm. Sci. 2017, 106 (7), 1722–1735. DOI:
(2) Brovč, E.V.; Mravljak, J.; Šink, R.; Pajk, S. Rational Design to Biologics Development: The Polysorbates Point of View. Int. J. Pharm. 2020, 581, 119285. DOI:
(3) Wuchner, K.; Yi, L.; Chery, C.; et al. Industry Perspective on the Use and Characterization of Polysorbates for Biopharmaceutical Products Part 1: Survey Report on Current State and Common Practices for Handling and Control of Polysorbates. J. Pharm Sci. 2022, 111 (5), 1280–1291. DOI:
(4) Wuchner, K.; Yi, L.; Chery, C.; et al. Industry Perspective on the Use and Characterization of Polysorbates for Biopharmaceutical Products Part 2: Survey Report on Control Strategy Preparing for the Future. J. Pharm Sci. 2022, 111 (11), 2955–2967. DOI:
(5) Kishore, R.S.K.; Kiese, S.; Fischer, S.; et al. The Degradation of Polysorbates 20 and 80 and its Potential Impact on the Stability of Biotherapeutics. Pharm. Res. 2011, 28 (5), 1194–1210. DOI:
(6) Dwivedi, M.; Blech, M.; Presser, I.; Garidel, P. Polysorbate Degradation in Biotherapeutic Formulations: Identification and Discussion of Current Root Causes. Int. J. Pharm. 2018, 552 (1–2), 422–436. DOI:
(7) Larson, N.R.; Wei, Y.; Prajapati, I.; et al. Comparison of Polysorbate 80 Hydrolysis and Oxidation on the Aggregation of a Monoclonal Antibody. J. Pharm. Sci. 2020, 109 (1), 633–639. DOI:
(8) Dwivedi, M.; Buske, J.; Haemmerling, F.; Blech, M.; Garidel, P. Acidic and Alkaline Hydrolysis of Polysorbates under Aqueous Conditions: Towards Understanding Polysorbate Degradation in Biopharmaceutical Formulations. Eur. J. Pharm. Sci. 2020, 144, 105211. DOI:
(9) Lippold, S.; Koshari, S.H.S.; Kopf, R.; et al. Impact of Mono- and Poly-Ester Fractions on Polysorbate Quantitation Using Mixed-Mode HPLC-CAD/ELSD and the Fluorescence Micelle Assay. J. Pharm. Biomed. Anal. 2017, 132 (5), 24–34. DOI:
(10) Brito, R.M.M.; Vaz, W.L.C. Determination of the Critical Micelle Concentration of Surfactants Using the Fluorescent Probe N-Phenyl-1-Naphthylamine. Anal. Biochem. 1986, 152 (2), 250–255. DOI:
(11) Martos, A.; Berger, M.; Kranz, W.; et al. Novel High-Throughput Assay for Polysorbate Quantification in Biopharmaceutical Products by Using the Fluorescent Dye DiI. J. Pharm. Sci. 2020, 109 (1), 646–655. DOI:
(12) Hewitt, D.; Zhang T.; Kao, Y.H. Quantitation of Polysorbate 20 in Protein Solutions Using Mixed Mode Chromatography and Evaporative Light Scattering Detection. J. Chromatogr. A 2008, 1215 (1–2), 156–160. DOI:
(13) Hewitt, D.; Alvarez, M.; Robinson, K.; et al. Mixed-Mode and Reversed-Phase Liquid Chromatography–Tandem Mass Spectrometry Methodologies to Study Composition and Base Hydrolysis of Polysorbate 20 and 80. J. Chromatogr. A 2011, 1218 (15), 2138–2145. DOI:
(14) Borisov, O.V.; Ji, J.A.; Wang, Y.J.; Vega, F.; Ling, V.T. Toward Understanding Molecular Heterogeneity of Polysorbates by Application of Liquid Chromatography–Mass Spectrometry with Computer-Aided Data Analysis. Anal. Chem. 2011, 83 (10), 3934–3942. DOI:
(15) Zhang, R.; Wang, Y.; Tan, L.; Zhang, H.Y.; Yang, M. Analysis of Polysorbate 80 and its Related Compounds by RP-HPLC with ELSD and MS Detection. J. Chromatogr. Sci. 2012, 50 (7), 598–607. DOI:
(16) Li, Y.; Hewitt, D.; Lentz, Y.K.; et al. Characterization and Stability Study of Polysorbate 20 in Therapeutic Monoclonal Antibody Formulation by Multidimensional Ultrahigh-Performance Liquid Chromatography–Charged Aerosol Detection–Mass Spectrometry. Anal. Chem. 2014, 86 (10), 5150–5157. DOI:
(17) Evers, D.H.; Schultz-Fademrecht, T.; Garidel, P.; Buske J. Development and Validation of a Selective Marker-Based Quantification of Polysorbate 20 in Biopharmaceutical Formulations Using UPLC QDa Detection. J. Chromatogr. B 2020, 1157, 122287. DOI:
(18) De Pra, M.; Ispan, D.A.; Lovejoy, K.; et al. Polysorbate 80 Profiling by HPLC with Mass and Charged Aerosol Detection. Thermo Fisher Scientific Application Note, 2021.
(19) Liu, H.; Jin, Y.; Menon, R.; et al. Characterization of Polysorbate 80 by Liquid Chromatography-Mass Spectrometry to Understand Its Susceptibility to Degradation and Its Oxidative Degradation Pathway. J. Pharm. Sci. 2022, 111, 323–334. DOI:
(20) Honemann, M.N.; Wendler, J.; Graf, T.; Bathke, A.; Bell, C.H. Monitoring Polysorbate Hydrolysis in Biopharmaceuticals Using a QC-Ready Free Fatty Acid Quantification Method. J. Chromatogr. B 2019, 1116, 1–8. DOI:
(21) Evers, D.H.; Carle, S.; Lakatos, D. Hydrolytic Polysorbate 20 Degradation - Sensitive Detection of Free Fatty Acids in Biopharmaceuticals via UPLC-QDa Analytics with Isolator Column. J. Chromatogr. B 2021, 1174, 122717. DOI:
(22) Vanhoenacker, G.; Steenbeke, M. ; Sandra, K.; Sandra, P. Profiling Nonionic Surfactants Applied in Pharmaceutical Formulations Using Comprehensive Two-Dimensional LC with ELSD and MS Detection. LCGC Eur. 2018, 31 (7), 360–370.
(23) David, F.; Ochiai N.; Sandra P. Stir Bar Sorptive Extraction: A Versatile, Sensitive and Robust Technique for Targeted and Untargeted Analyses. In Evolution of Solid Phase Microextraction Technology; RSC, 2023; pp. 175–213.
ABOUT THE AUTHORS
Piotr Alvarez is a senior scientist at RIC group (Kortrijk, Belgium).
Shauni Detremmerie is a scientist at RIC group.
Tatiana Cucu is group leader at RIC group.
Gerd Vanhoenacker is a senior scientist at RIC group.
Steffy Denorme is a senior research associate at RIC group.
Cindy Lecluyse is a senior research associate at RIC group.
Jorn Deryckere is a senior research associate at RIC group.
Bram D’Haenens is a senior research associate at RIC group.
Frank David is a principal scientist at RIC group.
Pat Sandra is the founder and advisor of the RIC group and emeritus professor at Ghent University (Ghent, Belgium).
ABOUT THE EDITOR
Koen Sandra is the editor of “Biopharmaceutical Perspectives”. He is CEO at RIC group and visiting professor at Ghent University. He is also a member of LCGC Europe’s editorial advisory board.
Articles in this issue
almost 3 years ago
GC Connections: Basic Care and Feeding of Your Detectoralmost 3 years ago
A Practical Understanding of Partition Coefficientsalmost 3 years ago
Events: ISSS 2023almost 3 years ago
Instrumental Innovations: July/August 2023