
Mass Spectrometry
Latest News

Latest Videos

More News

In this final part of our interview with Females in Mass Spectrometry (FeMS) Empowerment Award Winner Josephine Ouma, we discuss her future research efforts and what Women's History Month means to her.

In this second part of our interview with Females in Mass Spectrometry (FeMS) Empowerment Award Winner Josephine Ouma, we discuss her experience with mass spectrometry and how FeMS has impacted her life.

In this first part of our interview with Females in Mass Spectrometry (FeMS) Empowerment Award Winner Josephine Ouma, we discuss her PhD research and what inspired her to join FeMS.

The researchers explored ways to simplify retention time calculation across LC–MS and various other chromatographic methods.
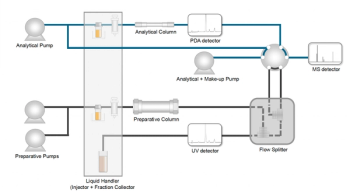
This article introduces a multi-step preparative purification workflow for synthetic peptides using liquid chromatography–mass spectrometry (LC–MS). The process involves optimizing separation conditions, scaling-up, fractionating, and confirming purity and recovery, using a single LC–MS system. High purity and recovery rates for synthetic peptides such as parathormone (PTH) are achieved. The method allows efficient purification and accurate confirmation of peptide synthesis and is suitable for handling complex preparative purification tasks.

Here is some of the most popular content posted on LCGC International this week.
A new method has been created for determining N-nitrosamines (NAs) in pharmaceutical preparations using liquid chromatography with tandem mass spectrometry.

A new approach to characterizing human milk oligosaccharides was developed using a combination of liquid chromatography, mass spectrometry, and other techniques.

New types of smart proteolytic samplers were tested for their effectiveness in liquid chromatography-mass spectrometry (LC–MS)-based protein quantification.

The benefits of nanobubbles in electrospray ionization mass spectrometry have been investigated.

Webinar Date/Time: Wed, Apr 16, 2025 11:00 AM EDT

Scientists from the Zhejiang Institute of Quality Scientists used liquid chromatography–tandem mass spectrometry to detect 35 PFAS in paper and plastic products.

Daisy Unsihuay Vila of Henry Ford Health (Detroit, Michigan) was awarded the 2025 Agilent Technologies and Females in Mass Spectrometry’s Darlene Solomon Award for her research focused on improving mass spectrometry (MS) techniques in clinical settings, particularly for drug monitoring and toxicology analysis.

Here is some of the most popular content posted on LCGC International this week.

In this interview, we spoke with Lee Ferguson of Duke University about his laboratory’s work accurately tracing these contaminants in complex environmental matrices.

During Waters Corporation’s 2025 Investor Day, the company’s top executives highlighted the company’s growth and hints at future points of development.

Here is some of the most popular content posted on LCGC International this week.

The company announced multiple new product releases including a series of compact gas chromatography systems, three UV-Vis spectrophotometers, a new line of triple quadrupole mass spectrometers, and more.

Webinar Date/Time: Thurs, March 27, 2025 11:00 AM EST

Here is some of the most popular content posted on LCGC International this week.

Guangdong University of Technology researchers used a gas chromatography-based method to detect nonylphenol isomers in sewage and wastewater sludge.
Scientists from Bristol Myers Squibb developed a method using supercritical fluid chromatography for chiral bioanalysis.

Here is some of the most popular content posted on LCGC International this week.


Thermal desorption and gas chromatography-tandem mass spectrometry were used to create a method for detecting airborne polycyclic aromatic hydrocarbon (PAH) samples.




