
A new way to control ion transports in microfluidic systems has the potential to improve the technology and expand applications.

A new way to control ion transports in microfluidic systems has the potential to improve the technology and expand applications.

A new differential ion mobility spectrometry device has been created that will greatly change how chemicals and substances are found and analyzed.

A recent study highlights the challenges and triumphs scientists studying charge determination mass spectrometry faced in the mid-1990s.

SIFT–MS analysis has potential to screen large numbers of packaging samples for the volatile MOH fraction, providing a rapid indication of packaging material contamination.

Pressure-enhanced liquid chromatography (PE-LC) offers a new approach for improving selectivity for large molecule separations. Examples shown here include short oligonucleotides in ion-pairing reversed-phase (IP-RP) liquid chromatography and larger nucleic acids in ion-exchange (IEX) chromatography.

Analysis of unidentified organofluorine compounds and known PFAS in environmental and human samples presents many challenges. Combustion ion chromatography is one technique that can help.

Using Earth as a figurative analogy, we explore the merits of flexibility when considering which liquid chromatography (LC) modality to use for any particular bioanalysis.

Interest in three-dimensional (3D) printing technology is expanding rapidly. What impact can it have in mass spectrometry? Robert Winkler, of the Center for Research and Advanced Studies Irapuato, in Guanajuato, Mexico, is exploring this question. We recently spoke with Prof. Winkler about this work.

Modern ion mobility–mass spectrometry (IM–MS) is a key separation technology for detailed molecular characterization studies and also as part of emerging data acquisition strategies for demanding small molecule and several applications. Here is what you need to know.

Using ion mobility, analytes that have the same molecular mass can be separated by their shape, centers of mass, and collision cross section, but challenges such as ion loss can still occur. A new development in ion mobility separation, high-resolution ion mobility (HRIM), addresses such problems, and is particularly well suited to challenging applications, such as glycosylation monitoring of biological drugs and vitamin D analysis.

Ion-exchange chromatography (IEX) is a frequently overlooked technique. This novel IEX method uses non-aqueous mobile phases to purify a non-crystalline intermediate after a reductive amination with D-xylose. IEX delivers a step change in strength and purity of the intermediate allowing successful downstream processing.


Sulphite is a preservative added to a vast range of foods and beverages to prevent browning or oxidation. Some individuals are sensitive to sulphite additives and may experience a range of allergic reactions. Therefore, both the U.S. Food and Drug Administration (FDA) and European Union (EU) laws require that the presence of sulphites be declared on food labels when the concentration exceeds 10 mg/L. Several analytical methods exist to measure sulphite in food and beverages, however, they suffer from repeatability issues, and can be quite cumbersome to perform. A patent has been filed for an innovative, fast, and accurate ion chromatographic (IC) method based on direct current (DC) mode electrochemical detection. This article explains more.

Characterization of mAbs and related products requires the identification of chromatographic peaks with MS. However, the conventional salt- and pH-gradient elution techniques used in IEX are inherently incompatible with MS. Ammonium acetate- and ammonium carbonate-based mobile phase systems have been recently applied in IEX-MS, but the influence of the eluent composition on peak shape and retention has not been discussed nor studied systematically until now. The aim of the present study was to understand the impact of ionic strength, buffer capacity, and pH-response on the retention behaviour and peak shape of mAb species.
Ion-pairing chromatography has traditionally been implemented with the addition of an ion-pairing reagent into the mobile phase. Here, an alternative method is presented, in which the ion-pairing reagent is deposited on column as a sample additive instead of in the mobile phase.
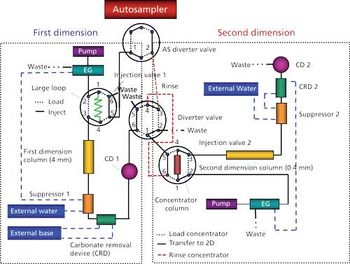
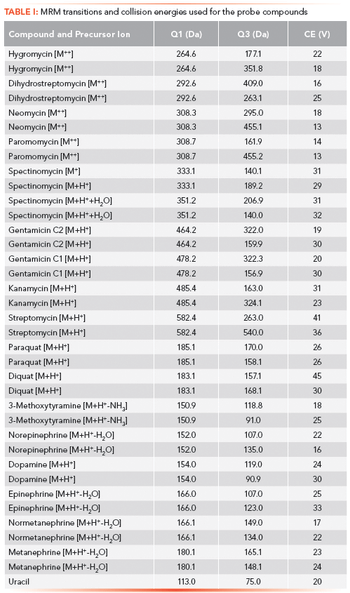
The disinfectants commonly used to treat public drinking water can react with naturally occurring organic and inorganic matter in the source water to form disinfection byproducts such as haloacetic acids. Here, we describe the use of two-dimensional matrix-elimination ion chromatography (MEIC) for haloacetic acid analysis. This method minimizes the impact of matrix ions.

Researchers from Zhejiang University, China, have created a graphene-coated polymeric stationary phase and demonstrated its performance separating inorganic anions, organic acids, carbohydrates, and amino acids using anion exchange chromatography.

In March 2015, the International Agency for Research on Cancer (IARC) published a report that stated that glyphosate was “probably carcinogenic to humans”. Ever since, the use of this chemical has been highly controversial. In some countries, including the USA and Australia, there are already limit values in effect for the weed killer.

Ion chromatography coupled to mass spectrometry (IC-MS) does not follow the same rules as coupling other modes of liquid chromatography to mass spectrometry (LC–MS). Leon Barron from the Analytical & Environmental Science Division, at King’s College London, UK, reveals some of the misconceptions surrounding IC and discusses contemporary trends and novel applications for this inventive technique.
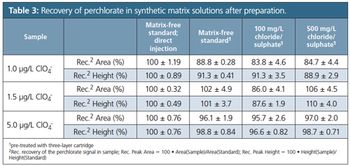
Perchlorate salts are relatively stable, soluble in water, and migrate into groundwater sources causing possible challenges for drinking water suppliers as dissolved perchlorate has been identified to impair normal thyroid function. The development of a sensitive analytical method for perchlorate determination is therefore important to protect public health. This article addresses the validation of ion chromatography with suppressed conductivity detection (IC–CD), applying isocratic elution to analyze perchlorate.





Analyzing Antarctic Ice Cores using Capillary Ion Chromatography