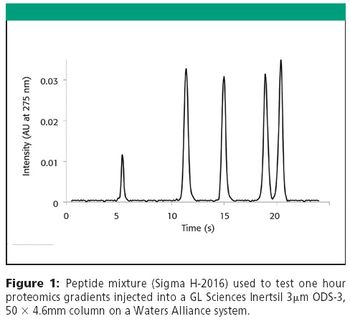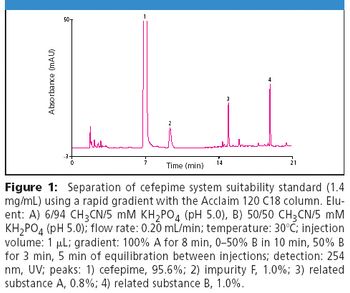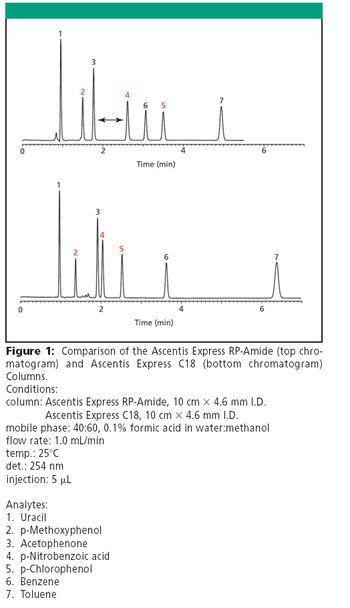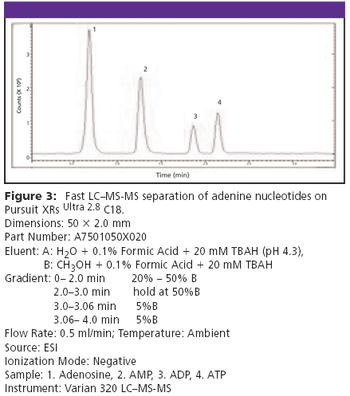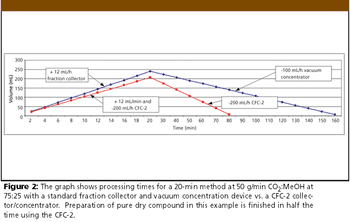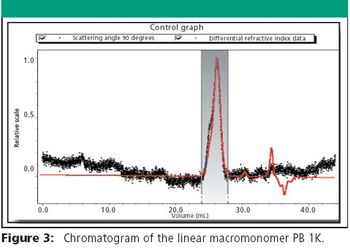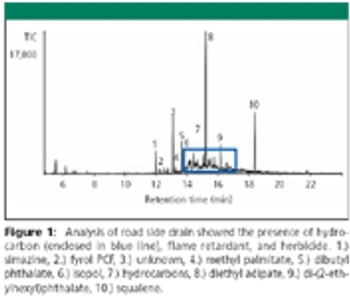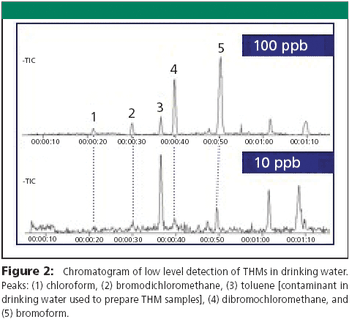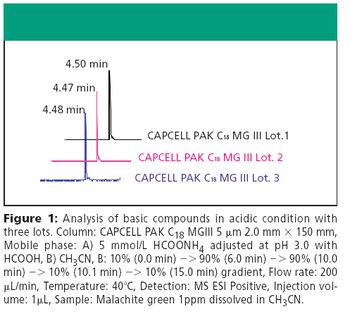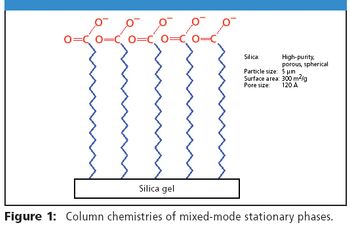
The Application Notebook
The separation and quantitation of drug substances and counterions are two important determinations in the pharmaceutical industry (1). Drug substances and counterions are determined by HPLC (often on reversed-phase columns) and by ion chromatography (IC), respectively. IC is the preferred method for selective and sensitive screening of both cationic and anionic pharmaceutical counterions (2). To increase the analysis throughput, it is desirable that for analysis of drug formulation both drug substance and counterions can be determined within a single run. Naproxen is a non-steroidal anti-inflammatory drug commonly used for treating moderate to severe pain, fever, inflammation, and stiffness. Naproxen sodium was approved by the U.S. Food and Drug Administration (FDA) as an over-the-counter drug. No reports were found describing one technique for the simultaneous determinations of both naproxen and the counterion (Na+).