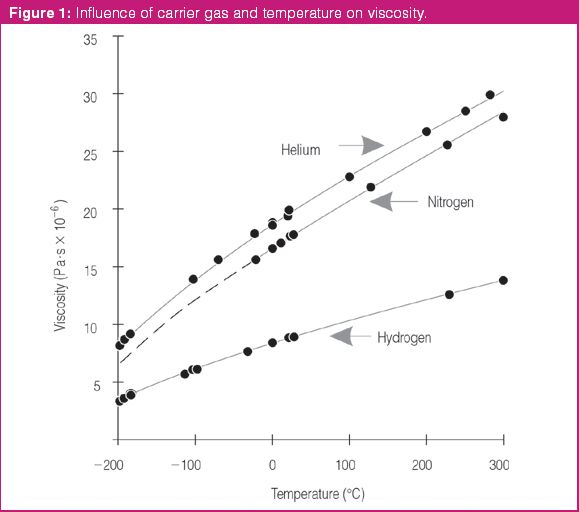
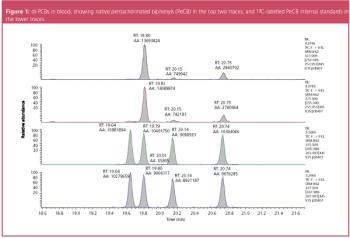
Melamine is an industrial chemical with a high nitrogen content that can cause kidney stones and lead to renal failure. In some instances, melamine has been added to baby formula and dairy products as a substitute for protein. This paper presents an efficient and definitive gas chromatography–mass spectrometry (GC–MS) method to identify melamine and related compounds based on the released US Food and Drug Administration (FDA) method.